 Evolution
Evolution
Nice Try! A Review of Alan Rogers’s The Evidence for Evolution
 I recently read The Evidence for Evolution by University of Utah professor of anthropology and biology Alan Rogers. The book is certainly concise, only 102 pages long. Christina Richards, of the University of South Florida, has praised it for presenting its arguments “in a respectful manner that is accessible to a broad audience without condescending language.” Indeed, I was pleasantly surprised to find that the book lives up to this claim.
I recently read The Evidence for Evolution by University of Utah professor of anthropology and biology Alan Rogers. The book is certainly concise, only 102 pages long. Christina Richards, of the University of South Florida, has praised it for presenting its arguments “in a respectful manner that is accessible to a broad audience without condescending language.” Indeed, I was pleasantly surprised to find that the book lives up to this claim.
Unlike Richard Dawkins in The Greatest Show on Earth or Jerry Coyne in Why Evolution is True, Alan Rogers refrains from sneering condescension and seeks to engage with the dissenting position respectfully and gracefully. For this, he is to be commended. What about the arguments presented in the book? The author attempts to demonstrate scientific support for the notion of common ancestry — drawing on several disciplines — as well as the proposition that an entirely unguided process (involving such mechanisms as natural selection and random mutation) can plausibly be responsible for what we find in biology.
In this review, I want to select some of what I consider to be the strongest argumentation presented.
The Mutability of Species
Chapter 1 (“Darwin’s Mockingbird”) and Chapter 2 (“Do Species Change?”) contain very little material that I would take issue with. The first chapter deals with island biogeography, particularly with respect to mockingbirds that Charles Darwin observed on the Galapagos islands, thus providing evidence for modest levels of common ancestry and even the ability of species to change and adapt to their environments. Since the Galapagos islands were populated exclusively by organisms that were best adapted for travel across large expanses of ocean, and the species found in the Galapagos islands were different from those found in South America, scientists justifiably concluded that those immigrants had subsequently changed following their arrival on the Galapagos. Rogers revisits island biogeography in Chapter 6 with a discussion of the Hawaiian flightless cricket Laupala. While I would be inclined to agree that island biogeography strongly suggests that different species of Laupala share a common ancestor, this is well within the bounds of what I understand to be the limits of evolution.
Chapter 2 deals with the question of whether species change. As with Chapter 1, this part of the book contains very little that I would disagree with. One example of evolutionary change given by Rogers is the emergence of methicillin-resistant Staphylococcus aureus (MRSA), which has acquired resistance to the antibiotics penicillin and methicillin. The mechanisms of such resistance, however, are less than impressive. Staphylococcus aureus acquires methicillin resistance by means of the mecA gene (Niemeyer et al., 1996), which sits on the mec operon. The mecA gene codes for a modified penicillin-binding protein (PBP2′), of high molecular weight, that possesses a decreased binding-affinity for β-lactams such as penicillins. In the presence of methicillin, PBP2′ takes over the functions of methicillin-sensitive PBPs and the bacterial peptidoglycan cell wall is structurally altered such that it has fewer oligomeric peptides. Since the mecA gene is located at the same chromosomal locus in all isolates of MRSA, it is probable that this adaptation occurred only a single time.
Rogers also mentions the recent resistance of MRSA to vancomycin, an antibiotic that inhibits bacterial cell wall biosynthesis.
You can visualize two of the main modes of vancomycin action, as well as one major bacterial mechanism of resistance, by looking at the following diagram (source):
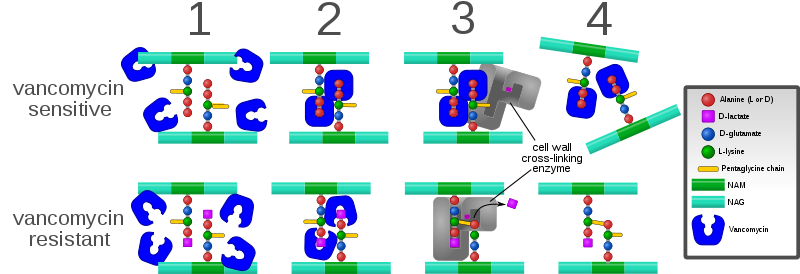
For sensitive strains, as seen in (1) and (2), the vancomycin targets the synthesis of the cell wall prior to cross-linking. The antibiotic binds to the two D-alanyl residues (D-Ala-D-Ala) that reside at the end of the polypeptides. This inhibits the formation of N-acetylmuramic acid (NAM) and N-acetylglucosamine (NAG), which normally comprise the cell wall backbone. In addition, as shown in (3) and (4), those polymers which do form are prevented from cross-linking. To achieve resistance, the c-terminal D-alanyl residue is substituted for D-lactate. This replaces the NH group with an oxygen to form an ester linkage. The replacement of the amide bond with an ester bond obviously carries consequences for the ability of the vancomycin to bind to the c-terminus of the stem peptide. This significantly reduces the potency of the antibiotic. This type of trivial change is, again, well within the bounds of what Michael Behe calls the edge of evolution.
Chapter 2 wraps up with a discussion of ring species, offered as evidence for the origin of new species by means of natural selection. As ENV recently discussed, however, such cases are often less than impressive.
The Fossil Record
Rogers’s book starts to get a little more controversial at Chapter 3, which asks the question “Does Evolution Make Big Changes?” The chapter begins with a discussion of the fossil record, detailing the whale fossil series, as well as the fish-to-amphibian transition.
Although on the face of it, the whale transition seems to be a relatively nice progression of forms, the main problem is that the transition takes place in far too narrow a window of time for it to be reasonably attributed to a Darwinian process.
The sheer force of this conundrum is only properly appreciated when one considers the multiple feats of anatomical novelty, innovative engineering and genetic rewiring necessary to change a terrestrial mammal like Pakicetus into a fully aquatic whale. Indeed, evolutionary biologist Richard Sternberg has argued that even many of the relatively minor changes are extremely unlikely to have occurred in the timeframe allowed. Consider the following small sample of necessary modifications:
- Counter-current heat exchanger for intra-abdominal testes
- Ball vertebra
- Tail flukes and musculature
- Blubber for temperature insulation
- Ability to drink sea water (reorganization of kidney tissues)
- Fetus in breech position (for labor underwater)
- Nurse young underwater (modified mammae)
- Forelimbs transformed into flippers
- Reduction of hindlimbs
- Reduction/loss of pelvis and sacral vertebrae
- Reorganization of the musculature for the reproductive organs
- Hydrodynamic properties of the skin
- Special lung surfactants
- Novel muscle systems for the blowhole
- Modification of the teeth
- Modification of the eye for underwater vision
- Emergence and expansion of the mandibular fat pad with complex lipid distribution
- Reorganization of skull bones and musculature
- Modification of the ear bones
- Decoupling of esophagus and trachea
- Synthesis and metabolism of isovaleric acid (toxic to terrestrial mammals)
- Emergence of blowhole musculature and neurological control
According to Sternberg’s calculations, and based on the equations of population genetics applied in a 2008 paper by Durrett and Schmidt in the Journal of Genetics, one may reasonably expect to see two coordinated mutations achieve fixation in the timeframe of around 43.3 million years. When one considers the magnitude of the engineering feat, such a scenario can only be ruled incredible. This problem is accentuated further when one considers that the majority of anatomical novelties unique to aquatic cetaceans (Pelagiceti) appeared during just a few million years — probably within 1-3 million years.
Until recently, the whale series, from Pakicetus (53 mya) to Dorudon and Basilosaurus (38 mya), has looked something like this:
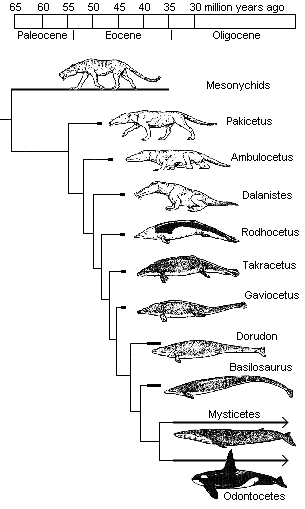
More recently, however, a jawbone was discovered that belonged to a fully aquatic whale dating to 49 million years ago, only four million years after Pakicetus! This means that the first fully aquatic whales date to around the time when walking whales (Ambulocetus) first appear. This substantially reduces the time window — to 4 or 5 million years, perhaps even less — that may be allotted to the Darwinian mechanism to accomplish truly radical engineering innovations and genetic rewiring. It also suggests that this fully aquatic whale existed before its previously assumed semi-aquatic archaeocetid ancestors.
Alan Rogers also discusses the widely known Tiktaalik fossil, the supposed intermediate between fish and amphibian tetrapods. This interpretation, however, has faced substantial problems since a study in Nature in 2010 reported the discovery of tetrapod footprints dated to 395 million years ago, substantially preceding Tiktaalik (which dates to some 375 million years ago). See this popular-level Nature news report for more.
Furthermore, as ENV’s Casey Luskin discusses here, evidence of a transition connecting the ray-finned fish-fin of Tiktaalik and the true tetrapod limbs of Acanthostega and Tulerpeton is sorely lacking.
No mention is given in Rogers’s book of the Cambrian radiation of animal life, which occurred some 540 million years ago — a dramatic explosion substantially undercutting the notion of universal common descent.
Traces of Common Descent in Living Organisms?
Alan Rogers’s next focus is on the “traces of common descent in living organisms.” Although there are several different classes of this type of molecular evidence, Rogers discusses the distribution of transposons, which he claims are “worse than useless” and which “consist of molecular parasites” that “are shortish stretches of DNA that do nothing useful for you and are good at just one thing. They have evolved the ability to hijack the machinery of your cell, tricking it into copying the parasite and inserting the copy into some random spot in your DNA.” Contrary to Rogers’s claim here, functional utility has been identified in the case of many transposons. For a recent review on Alu, the most common form of human transposon, see this paper by Pandey and Murkerji (2011) entitled “From ‘JUNK’ to Just Unexplored Noncoding Knowledge: The Case of Transcribed Alus.”
Rogers draws on two arguments from improbability: (1) the low likelihood of a transposon insert being present at the same site (and with the same orientation) across multiple taxa except by common descent; and (2) the low likelihood of the distribution of shared transposons forming a nested hierarchy that resembles a phylogenetic tree except by common descent. The examples he gives are (1) the phylogenetic relationship between bottlenose dolphins, narwhales, sperm whales and humpback whales; and (2) the phylogenetic relationship among deer, cows, hippopotamuses, humpback whales and beaked whales. While few would be surprised to learn that species of the mammalian order cetaceans (whales, dolphins, porpoises) are related by descent, I am not convinced that transposons shared between multiple taxa always indicate descent from a common ancestor.
According to Levy et al. (2009),
Throughout evolution, eukaryotic genomes have been invaded by transposable elements (TEs). Little is known about the factors leading to genomic proliferation of TEs, their preferred integration sites and the molecular mechanisms underlying their insertion. We analyzed hundreds of thousands nested TEs in the human genome, i.e. insertions of TEs into existing ones. We first discovered that most TEs insert within specific ‘hotspots’ along the targeted TE. In particular, retrotransposed Alu elements contain a non-canonical single nucleotide hotspot for insertion of other Alu sequences. We next devised a method for identification of integration sequence motifs of inserted TEs that are conserved within the targeted TEs. This method revealed novel sequences motifs characterizing insertions of various important TE families: Alu, hAT, ERV1 and MaLR. Finally, we performed a global assessment to determine the extent to which young TEs tend to nest within older transposed elements and identified a 4-fold higher tendency of TEs to insert into existing TEs than to insert within non-TE intergenic regions. Our analysis demonstrates that TEs are highly biased to insert within certain TEs, in specific orientations and within specific targeted TE positions. TE nesting events also reveal new characteristics of the molecular mechanisms underlying transposition. [emphasis added]
The researchers documented that these transposable elements routinely preferentially insert into certain classes of already-present transposable elements, and do so with a specific orientation and at specific locations within the mobile element sequence.
Another paper by Li et al. (2009), appearing in Science at around the same time, documented that in the water flea genome, introns appear to have been integrated repeatedly into the same loci in different genomes. This led the acclaimed evolutionary biologist Michael Lynch to note,
Remarkably, we have found many cases of parallel intron gains at essentially the same sites in independent genotypes. This strongly argues against the common assumption that when two species share introns at the same site, it is always due to inheritance from a common ancestor.
A recent study by Spradling et al. (2011) documented Drosophila P elements preferentially transpose to replication origins. They report,
P element insertions preferentially target the promoters of a subset of genes, but why these sites are hotspots remains unknown. We show that P elements selectively target sites that in tissue-culture cells bind origin recognition complex proteins and function as replication origins.
I would tend to agree with Alan Rogers, however, that the nested hierarchical pattern of distribution is what one would expect to observe given the assumption of common ancestry. I also agree that the evidence seems to indicate that these elements are indeed inserts into the sites that they presently occupy. This is evidenced by the presence of the telltale target site duplication, which results from the staggered cut induced by the transposase enzyme (which is subsequently filled in by the DNA polymerase) and its repair by the DNA ligase (which closes the sugar-phosphate backbone). Transposon insertion is thus signposted by short direct repeats followed by inverted repeats.
Can Evolution Explain Design?
Chapter 4 is primarily concerned with the plausibility of a complex eye having evolved by natural selection. Rogers presents the familiar argument that the eye began as a light-sensitive spot that was modified into a shallow cup in order to allow it to be sensitive only to light straight ahead (enabling the organism to find food more easily). According to this model, the eye pit subsequently deepened, thus improving its directional capability. Rogers explains that the eye “has become a pinhole camera, forming an image on the eye spot (which we can now call a retina). Not only can this creature sense light, it can really see.” The next stage is for the eye to secrete transparent mucus (“vitreous mass”) to form a lens, allowing the rays of light to bend toward the center. The gradual improvement of the eye continues in this manner to form a complex camera eye, such as we find in vertebrates today. This argument claims traction from the fact that all of those intermediate forms of eye are found in creatures alive today.
Just as common evolutionary explanations for the origin of the bacterial flagellum fundamentally trivialize the sheer engineering sophistication and complexity of the flagellum, so too does this evolutionary scenario trivialize the sheer engineering sophistication of the eye. It also ignores the molecular detail of the biochemical mechanisms of vision (which Michael Behe discusses here). In his review of Richard Dawkins’s Climbing Mount Improbable, David Berlinski has this to say:
In fact, the difficulties are very considerable. A single retinal cell of the human eye consists of a nucleus, a mitochondrial rod, and a rectangular array containing discrete layers of photon-trapping pigment. The evolutionary development of the eye evidently required an increase in such layers. An inferential staircase being required, the thing virtually constructs itself, Dawkins believes, one layer at a time. “The point,” he writes, “is that ninety-one membranes are more effective…than ninety, ninety are more effective that eighty-nine, and so on back to one membrane, which is more effective than zero.”
This is a plausible scheme only because Dawkins has considered a single feature of the eye in isolation. The parts of a complex artifact or object typically gain their usefulness as an ensemble. A Dixie Cup consists of a tube joined to a disk. Without the disk, the cup does not hold less water than it might; it cannot hold water at all. And ditto for the tube, the two items, disk and tube, forming an irreducibly complex system.
What holds for the Dixie Cup holds for the eye as well. Light strikes the eye in the form of photons, but the optic nerve conveys electrical impulses to the brain. Acting as a sophisticated transducer, the eye must mediate between two different physical signals. The retinal cells that figure in Dawkins’ account are connected to horizontal cells; these shuttle information laterally between photoreceptors in order to smooth the visual signal. Amacrine cells act to filter the signal. Bipolar cells convey visual information further to ganglion cells, which in turn conduct information to the optic nerve. The system gives every indication of being tightly integrated, its parts mutually dependent.The very problem that Darwin’s theory was designed to evade now reappears. Like vibrations passing through a spider’s web, changes to any part of the eye, if they are to improve vision, must bring about changes throughout the optical system. Without a correlative increase in the size and complexity of the optic nerve, an increase in the number of photoreceptive membranes can have no effect. A change in the optic nerve must in turn induce corresponding neurological changes in the brain. If these changes come about simultaneously, it makes no sense to talk of a gradual ascent of Mount Improbable. If they do not come about simultaneously, it is not clear why they should come about at all.
The same problem reappears at the level of biochemistry. Dawkins has framed his discussion in terms of gross anatomy. Each anatomical change that he describes requires a number of coordinate biochemical steps. “[T]he anatomical steps and structures that Darwin thought were so simple,” the biochemist Mike Behe remarks in a provocative new book (Darwin’s Black Box), “actually involve staggeringly complicated biochemical processes.” A number of separate biochemical events are required simply to begin the process of curving a layer of proteins to form a lens. What initiates the sequence? How is it coordinated? And how controlled? On these absolutely fundamental matters, Dawkins has nothing whatsoever to say.
Alan Rogers also makes mention of Dan-Eric Nilsson and Susanne Pelger’s famous work on the evolution of the eye. This research, however, made a number of crucial assumptions that bear strongly on their results. Indeed, the failing of their study becomes strikingly evident from the word go when they state that “Taking a patch of pigmented light-sensitive epithelium as the starting point, we avoid the more inaccessible problem of photoreceptor cell evolution.” This is a remarkable admission on their part. The primary problem, I think, with their paper — and indeed with the simplistic scenario we have just discussed — relates to their depiction of substantial morphological innovation arising by means of evolutionary pressures without any regard given to the biochemical difficulties of actualizing those changes.
The selective pressure in Nilsson and Pelger’s study relates to the eye’s visual acuity: that is, its efficacy in distinguishing between different parts of an image (which requires that different photoreceptors possess different fields of view).
One important assumption they make is that modification of shape (e.g. making the eye more concave) is based on genes and is thus heritable. Although their model assumes that such morphological variations are only 50% hereditable, the critical assumption is that they are heritable at all. They also assume that, irrespective of how much morphological change has been accomplished in the past, more is indefinitely possible. This is an important premise because evolution by recombination of existing genes is very limited — it thus entails the origin of novel genetic material.
But let’s briefly consider Nilsson and Pelger’s calculation concerning the speed of eye evolution. The size of evolutionary steps is quantified in terms of visual acuity. The value they adopt for enhancement of visual acuity is 1%. As they themselves highlight, however, as performance increases, so too does the magnitude of each improvement, in addition to the size of morphological change driving each improvement. This is a major problem with their approach — basing their scenario on performance rather than step changes of structure — and somewhat undermines our confidence in their claim, stated in the title of the paper, that their study represents “a pessimistic estimate of the time required for an eye to evolve.”
For further discussion of Nilsson and Pelger, see David Berlinski’s article here.
Alan Rogers further claims that there are “traces of common descent” in opsins, in developmental genes and in lens proteins. This argument also fails, however, since there are common genes found in different taxa that cannot be accounted for in terms of common descent.
Consider, for example, rhodopsin, the key molecule involved in absorbing light across all modern phyla. This is true not only of the eukaryotes, but also of photosensitive prokaryotes such as some bacteria and algae. It is not the case, however, that the molecule in all these groups is derivative from a common photosensitive ancestor. In their classic work (Salvini-Plawen LV, Mayr E On the evolution of photoreceptors and eyes. Evol. Biol. 1977;10:207-263), Mayr and Salvini-Plawen note,
All the evidence however indicates that the earliest invertebrates, or at least those that gave rise to the more advanced phyletic lines, had no photoreceptors.
In fact, this conclusion was evident from a study of the molluscs alone:
The molluscs display greatest diversity in the differentiation of eyes among all groups of animals and 7-11 different lines can be distinguished; the ancestral stock was obviously devoid of photoreceptors and neither [various mollusc groups] nor most original larvae […] possess photoreceptors.
Or consider equivalent homeobox genes that play analogous roles in development across taxa where no common ancestor is to be found. In species as different as insects and mammals (which possess compound and camera eyes respectively) — the common ancestor of which, according to evolutionary reckoning, lived so long ago that it did not have eyes — the embryological formation of the eyes uses remarkably similar genes (e.g., eyeless and Pax6 respectively).
Not only is Pax6 common to the development of vertebrate and invertebrate eyes, but so too are quite a number of other transcription factors. Among these are the mammalian “Six” genes, and their analogue in Drosophila called sine oculis. These genes are deployed somewhat later in development than is Pax6. Even the convergently deployed genes Dach and dac (vertebrates and Drosophila respectively) are seemingly homologous in structure, but each possesses quite a restricted role and is utilized late in development. Moreover, the eyes of vertebrates develop from two embryonic tissues, namely the epithelium and optic vesicle, whereas the eyes of Drosophila develop from a single embryonic tissue: the imaginal disc.
Rogers concludes this chapter by claiming that, just as the apparent irreducible complexity of the eye has now been shown to be no such thing, so too has Michael Behe’s claim that the bacterial flagellum represents an irreducibly complex system. In support of this, he notes that,
It turns out that a simpler organ, involving several of the same proteins, is used by some bacteria to inject toxins into other cells. It is simply not true that these proteins have no function apart from their role in the flagellum.
This argument has been refuted so thoroughly on this site (e.g. here) and elsewhere (e.g. here) that there is no need to do so again now.
What About Suboptimal Design?
Chapter 5 of the book is entitled “Peaks and Valleys,” and highlights the fact that a population may “get stuck on inferior fitness peaks even when a superior peak is (so to speak) in plain view.” This may occur when a maladaptive valley exists between one fitness peak and another. This means that evolution often produces suboptimal design, unable to correct it without passing through maladaptive intermediates — Rogers provides the example of the circuitous route taken by the vas deferens around the ureter, the recurrent laryngeal nerve, and the vertebrate eye.
In the case of the route taken by the vas deferens, the testes develop from the same structure as the ovaries in females, which is called the genital ridge and which is near where the kidneys will develop. There is a cord called the gubernaculum testis, which connects the testis to the scrotum. As the fetus grows, the gubernaculum testis does not, and thus the testis is pulled forward, eventually through the body wall and into the scrotum. The lengthening vas deferens merely follows. Furthermore, before the vas deferens joins the urethra, there needs to be a place where the seminal vesicle adds its contents.
As for the recurrent laryngeal nerve, ENV’s Casey Luskin notes several potential design reasons for the chosen route:
(1) There is evidence that supposed fundamental evolutionary constraints which would prevent loss of the circuitous route of the RLN do not exist. This implies that there is some beneficial function for the circuitous route.
(2) The path of the RLN allows it to give off filaments to the heart, to the mucous membranes and to the muscles of the trachea along the way to the larynx.
(3) There is dual-innervation of the larynx from the SLN and RLN, and in fact the SLN innervates the larynx directly from the brain. The direct innervation of the larynx via the superior laryngeal SLN shows the laryngeal innervations in fact follows the very design demanded by ID critics like Jerry Coyne and Richard Dawkins. Various medical conditions encountered when either the SLN or RLN are damaged point to special functions for each nerve, indicating that the RLN has a specific laryngeal function when everything is functioning properly. This segregation may be necessary to achieve this function, and the redundancy seems to preserve some level of functionality if one nerve gets damaged. This dual-innervation seems like rational design principle.
Luskin also quotes Michael Egnor, who lists some other interesting advantages to the route taken by the RLN:
There is actually a design advantage to the course of the recurrent nerves, if one wishes to pursue this line of argumentation. The course of the nerves brings them through the mediastinum, where the heart and lungs meet. There are many lymph nodes there, and enlargement of these lymph nodes from processes such as cancer or infection (e.g., tuberculosis) often irritates these nerves and causes hoarseness or coughing. The course of the nerves reveals disease in an otherwise hidden part of the body (deep in the chest) by interfering with a process (speech) that is readily evident. It serves as an early warning to get medical care (or, with infectious diseases, as a warning to others that this person is ill), and this early warning has saved many more lives than the redundant course of the nerves has cost lives. The risk/benefit ratio needs to be examined comprehensively before one claims that the course of the nerves is biologically disadvantageous.
Egnor further notes,
Of course ID advocates have never claimed perfect design. But the argument that the anatomy of the recurrent laryngeal nerve is evidence for “bad design” fails on many levels. The descent of the recurrent nerves below the aortic arch and subclavian artery is the result of patterns of coalescence and movements of components of the aortic arch during embryogenesis. It appears that proximity of various layers and structures in the embryo serve to guide embryogenesis (it’s called induction). The details of this process are only beginning to be understood, and the Darwinist argument that the relationship between the recurrent nerves and the aortic arch is evidence of bad design fails to take into account the enormous complexities of embryonic development. It’s analogous to a 3 year old taking apart a computer and asserting that it was designed badly because some of the circuit board patterns were “curvy” instead of straight. The design wisdom of the anatomy of the recurrent nerves can only be judged by someone who knows all of the design specifications necessary for that region of the human body. Even the best embryologists are preschoolers when it comes to that.
On the design of the eye, retinal cells require a very large oxygen supply, and hence a very large blood supply. Blood cells absorb light. If blood cells invade retinal cells, the consequence can be blindness. This entails that the retinal cells need to receive a blood supply from vessels that do not block the light. Since red blood cells readily absorb light, this demand requires that the retina be wired in the manner in which it is. As is often pointed out, squids and octopuses have “correctly” wired retinas that face outward. But these organisms are exothermic — they do not require the same blood supply to the retina.
In any case, as Luskin explains here, undesirable intelligent design is still design! Since intelligent design, unlike natural theology, does not make claims concerning the identity, attributes or nature of the designer, ID makes no claims about the optimality of design. Human engineers — like those Microsoft — design suboptimal systems all the time.
Has There Been Enough Time?
Chapter 7 asks the question “Has there been enough time?” This chapter is taken up with dispelling popular notions that the earth is young (thousands of years old as opposed to billions). Since I fully agree that the earth is very ancient (some 4.6 billion years old), there is not much to disagree with. Alan Rogers does not, however, even attempt to demonstrate that the conventional age of the earth offers sufficient time to the evolutionary process. Indeed, papers like this one and this one suggest that this is not the case even for relatively “trivial” changes.
Did Humans Evolve?
The last two chapters of the book concern human evolution. Chapter 8 asks “Did humans evolve?” and Chapter 9 asks “Are we still evolving?” Alan Rogers discusses both fossil and genetic evidence for human descent from other primates. On the fossil evidence, Rogers writes,
[S]kulls older than 2 million years are called australopithecines and those younger than 1.8 million years are seen as members of our own genus, Homo. In between lies a troublesome group…that has proved difficult to name. They have traditionally been called Homo habilis, but this name has never seemed entirely satisfactory. Some authors see these creatures as australopithecines; others see them as bona fide members of Homo.
Lately, the consensus has swung toward the view that there are really two species here, H. habilis and another called H. rudolfensis. But that has not worked out too well either. As paleoanthropologist Ian Tattersall recently remarked, “just what — if anything — Homo rudolfensis is remains anybody’s guess”. It is interesting that these fossils are so hard to classify. In some features, they resemble the earlier forms. In others, they resemble later ones. They are, in other words, intermediate.
As Casey Luskin discusses here, here and here, however, there is good reason to question whether these fossils really do represent intermediate links.
Rogers also discusses the evidence from genetics, including the evidence from transposons and from pseudogenes such as urate oxidase (which, in most mammals, is involved in getting rid of uric acid) and GULO (which encodes the final enzyme in the ascorbic acid biosynthesis pathway). Rogers explains — and with this I would agree — how the phenomenon of incomplete lineage sorting can result in phylogenetic incongruity between closely related groups. Incomplete lineage sorting occurs when a second split occurs after an initial split but before genetic material in the two daughter species has been fully sorted — this is followed by each of the three species losing a random gene variant by genetic drift. This should give ID proponents cause for caution before asserting that incongruence between very closely related groups necessarily undermines common ancestry.
In the case of pseudogenes, there is often evidence of function. For a recent review, see this 2011 paper by Pink et al. (“Pseudogenes: Pseudo-functional or key regulators in health and disease?”). In fact, pseudogene similarities are often taken as evidence for functional constraint (Balakirev and Ayala, 2003; Khachane and Harrison, 2009). See my article here for further discussion of the GULO pseudogene.
As previously indicated, there is evidence to suggest that transposons are genuine inserts, and that they may have quite specific target-site preferences (though there is still the nested hierarchical pattern of distribution to contend with). As Ann Gauger explains here, however, there are often cases where shared morphological, structural or sequence similarities cannot be explained by common ancestry. This is true even within primates (e.g. Kriener et al., 2000; O’Huigin, 1995). For further discussion, see Casey Luskin’s “Primer on the Tree of Life”.
Conclusion
Alan Rogers deserves praise for his civil and respectful tone. Unlike many other such works, his book does not resort to condescending derision and rhetoric. However, it does not present anything fundamentally new, no arguments or evidence that hasn’t been addressed in detail before. Although some evidence is presented for fairly modest levels of common ancestry, the grander claim of universal common ancestry is not justified, nor does the author impart confidence in the adequacy of the neo-Darwinian mechanism.
