 Evolution
Evolution
Jerry Coyne’s Chapter on the Fossil Record Fails to Show “Why Evolution Is True”
In my article yesterday, I began to review Jerry Coyne’s book Why Evolution is True (2009). In this second installment, I offer a critical analysis of Chapter 2 of Coyne’s book.
Chapter 2: Written in the Rocks
Chapter 2 addresses the fossil evidence for common descent. The first part of the chapter I generally agree with. Coyne provides a summary outline of the fossilization process and an introduction to radioisotope dating methods for estimating the age of rocks (radiometric dating of meteorites also allows us to estimate the age of our solar system as being approximately 4.6 billion years old).
I agree with Coyne that the fossil record generally exhibits a trend of simpler organisms preceding more complex ones (for example, bacteria roamed the planet exclusively for the first two billion years of life on earth). This is, for sure, what would be expected given common ancestry. One feature of the fossil record that would not be expected under Darwin’s theory, however, is the observed pattern of morphological disparity preceding diversity. Taxonomists classify organisms into categories: species are the very lowest taxonomic category. Species are classified into different genera. Genera are classified into different families. Families are classified into different orders. Orders are classified into different classes. And classes are classified into different phyla. Phyla are among the very highest taxonomic categories (only kingdom and domain are higher), and correspond to the high level of morphological disparity that exists between different animal body plans. Phyla include such groupings as chordates, arthropods, mollusks, and echinoderms.
Darwin’s theory would predict a cone of diversity whereby the major body-plan differences (morphological disparity) would only appear in the fossil record following numerous lower-level speciation events. What is interesting about the fossil record is that it shows the appearance of the higher taxonomic categories first (virtually all of the major skeletonized phyla appear in the Cambrian, with no obvious fossil transitional precursors, within a relatively small span of geological time). As Roger Lewin (1988) explains in Science,
Several possible patterns exist for the establishment of higher taxa, the two most obvious of which are the bottom-up and the top-down approaches. In the first, evolutionary novelties emerge, bit by bit. The Cambrian explosion appears to conform to the second pattern, the top-down effect.
Erwin et al. (1987), in their study of marine invertebrates, similarly conclude that,
The fossil record suggests that the major pulse of diversification of phyla occurs before that of classes, classes before that of orders, orders before that of families. The higher taxa do not seem to have diverged through an accumulation of lower taxa.
Indeed, the existence of numerous small and soft-bodied animals in the Precambrian strata undermines one of the most popular responses that these missing transitions can be accounted for by them being too small and too-soft bodied to be preserved. See my article on the Cambrian explosion here for responses to other popular objections.
Coyne covers three major evolutionary transitions purportedly documented by the fossil record. These include the evolution of amphibians from fish, the evolution of birds from reptiles, and the evolution of whales from terrestrial mammals. But just how convincing are these examples?
Onto the Land: From Fish to Amphibians
Coyne commences his treatment of the fossil record with a discussion of the fish-to-amphibian transition. He explains,
One of the greatest fulfilled predictions of evolutionary biology is the discovery, in 2004, of a transitional form between fish and amphibians. This is the fossil species Tiktaalik roseae, which tells us a lot about how vertebrates came to live on the land. Its discovery is a stunning vindication of the theory of evolution.
Until about 390 million years ago, the only vertebrates were fish. But, thirty million years later, we find creatures that are clearly tetrapods: four-footed vertebrates that walked on land. These early tetrapods were like modern amphibians in several ways: they had flat heads and bodies, a distinct neck, and well-developed legs and limb girdles. Yet they also show strong links with earlier fishes, particularly the group called “lobe-finned fishes,” so called because of their large body fins that enabled them to prop themselves up on the bottom of shallow lakes or streams. The fish-like structures of early tetrapods include scales, limb bones, and head bones. (pp. 35-36)
According to Coyne, the evolution of amphibians from fish makes a testable prediction. He notes,
If there were lobe-finned fishes but no terrestrial vertebrates 390 million years ago, and clearly terrestrial vertebrates 360 million years ago, where would you expect to find the transitional forms? Somewhere in between. Following this logic, [Neil] Shubin predicted that if transitional forms existed, their fossils would be found in strata around 375 million years old. Moreover, the rocks would have to be from freshwater rather than marine sediments, because late lobe-finned fish and early amphibians both lived in fresh water.
Searching his college geology textbook for a map of exposed freshwater sediments of the right age, Shubin and his colleagues zeroed in on a paleontologically unexplored region of the Canadian Arctic: Ellesmere Island, which sits in the Arctic Ocean north of Canada. And after five long years of fruitless and expensive searching, they finally hit pay dirt: a group of fossil skeletons stacked atop one another in sedimentary rock from an ancient stream. When Shubin first saw the fossil face poking out of the rock, he knew that he had at last found his transitional form. In honor of the local Inuit people and the donor who helped fund the expeditions, the fossil was named Tiktaalik roseae (“Tiktaalik” means “large freshwater fish” in Inuit, and “roseae” is a cryptic reference to the anonymous donor). (p. 37)
 The problem with Neil Shubin’s prediction, described by Coyne, is that tetrapod tracks have been discovered that date to 397 million years ago (Nied?wiedzki et al., 2010) — that’s some 20 million years before Tiktaalik! As a Nature article put it,
The problem with Neil Shubin’s prediction, described by Coyne, is that tetrapod tracks have been discovered that date to 397 million years ago (Nied?wiedzki et al., 2010) — that’s some 20 million years before Tiktaalik! As a Nature article put it,
There was a consensus that the divergence between some elpistostegalians (such as Tiktaalik or Panderichthys) and tetrapods might have occurred during the Givetian, 391-385 Myr ago. Coeval with the earliest fossil tetrapods, trackways dating to the Late Devonian were evidence for their ability to walk or crawl on shores.
Now, however, Nied?wiedzki et al. lob a grenade into that picture. They report the stunning discovery of tetrapod trackways with distinct digit imprints from Zache?mie, Poland, that are unambiguously dated to the lowermost Eifelian (397 Myr ago). This site (an old quarry) has yielded a dozen trackways made by several individuals that ranged from about 0.5 to 2.5 metres in total length, and numerous isolated footprints found on fragments of scree. The tracks predate the oldest tetrapod skeletal remains by 18 Myr and, more surprisingly, the earliest elpistostegalian fishes by about 10 Myr.
Coyne notes the “features that make [Tiktaalik] a direct link between the earlier lobe-finned fish and the later amphibians.” He explains,
With gills, scales, and fins, it was clearly a fish that lived its life in water. But it also has amphibian-like features. For one thing, its head is flattened like that of a salamander, with the eyes and nostrils on top rather than on the sides of the skull. This suggests that it lived in shallow water and could peer, and probably breathe above the surface. The fins had become more robust, allowing the animal to flex itself upward to help survey its surroundings. And, like the early amphibians, Tiktaalik has a neck. Fish don’t have necks — their skull joins directly to their shoulders.
Most importantly, Tiktaalik has two novel traits that were to prove useful in helping its descendants invade the land. The first is a set of sturdy ribs that helped the animal pump air into its lungs and move oxygen from its gills (Tiktaalik could breathe both ways). And instead of the many tiny bones in the fins of lobe-finned fish, Tiktaalik had fewer and sturdier bones in the limbs — bones similar in number and position to those of every land creature that came later, including ourselves. In facts, its limbs are best described as part fin, part leg. (pp. 37-38)
But do these supposed intermediate features necessarily imply Tiktaalik‘s transitional status? After all, a modern species of African lungfish (Protopterus annectens) “uses a range of pelvic fin-driven, tetrapod-like gaits, including walking and bounding, in an aquatic environment, despite having a derived limb endoskeleton and primitively small, muscularly supported pelvis,” and even “lifts its body clear of the substrate using its pelvic fins” (King et al., 2011). But no one thinks that this species is an intermediate link connecting fish and tetrapod amphibians. Similarly, the walking catfish (Clarias batrachus) is also an air-breathing fish that is able to walk across dry land, and keep itself upright using its pectoral fins.
The following points are worth bearing in mind. First, whereas fish (including Tiktaalik) possess small pelvic fins relative to the pectoral fins, tetrapod hind limbs are uniformly more robust than the forelimbs. Secondly, Tiktaalik has no true toe or finger bones. Instead, like all other fish, it possesses bony fin rays (which can hardly be considered homologous to digits). As ENV’s Casey Luskin discusses here, there is a large unbridged gap between “the ray-finned fish-fin of Tiktaalik and the true tetrapod limbs of Acanthostega and Tulerpeton.”
Into Thin Air: The Origin of Birds
 The second of Coyne’s examples is the evolution of birds from theropod dinosaurs, and for this he offers a discussion of Archaeopteryx, in addition to other examples of purported transitional fossils (e.g., Sinornithosaurus millenii). Features that have convinced many paleontologists of Archaeopteryx‘s transitional status with respect to theropod dinosaurs and birds include the fact that it possesses fully developed feathers (including primaries and secondaries) which were arranged on its wings in a manner similar to modern birds, as well as its reptilian features such as a long bony tail, the claws on its forelimbs and hindlimbs, and its teeth. Other fossilized birds, however, have been found to have teeth (e.g., Hesperornis) and, indeed, some modern birds (e.g., the ostrich) possess fingers on their wings. Moreover, an interesting paper appeared in the Journal of Morphology in 2005, challenging the evidence for feathered dinosaurs (Fedducia et al., 2005). The authors conclude that,
The second of Coyne’s examples is the evolution of birds from theropod dinosaurs, and for this he offers a discussion of Archaeopteryx, in addition to other examples of purported transitional fossils (e.g., Sinornithosaurus millenii). Features that have convinced many paleontologists of Archaeopteryx‘s transitional status with respect to theropod dinosaurs and birds include the fact that it possesses fully developed feathers (including primaries and secondaries) which were arranged on its wings in a manner similar to modern birds, as well as its reptilian features such as a long bony tail, the claws on its forelimbs and hindlimbs, and its teeth. Other fossilized birds, however, have been found to have teeth (e.g., Hesperornis) and, indeed, some modern birds (e.g., the ostrich) possess fingers on their wings. Moreover, an interesting paper appeared in the Journal of Morphology in 2005, challenging the evidence for feathered dinosaurs (Fedducia et al., 2005). The authors conclude that,
Our ?ndings show no evidence for the existence of protofeathers and consequently no evidence in support of the follicular theory of the morphogenesis of the feather. Rather, based on histological studies of the integument of modern reptiles, which show complex patterns of the collagen ?bers of the dermis, we conclude that “protofeathers” are probably the remains of collagenous ?ber “meshworks” that reinforced the dinosaur integument. These “meshworks” of the skin frequently formed aberrant patterns resembling feathers as a consequence of decomposition.
The researchers also “examine evidence relating to the most critical character thought to link birds to derived theropods, a tridactyl hand composed of digits 1-2-3.” They find that “the evidence supports interpretation of bird wing digit identity as 2,3,4, which appears different from that in theropod dinosaurs.”
Another paper published in the Journal of Morphology raises a further problem for the evolution of birds from theropod dinosaurs (Quick and Ruben, 2009). The paper reports,
Although crocodilian lung and cardiovascular organs are markedly less specialized than the avian heart and lung air-sac system, all living archosaurs possess four-chambered hearts and heterogeneously vascularized, faveolar lungs. In birds, normal lung function requires extensive, dorsally situated nonvascularized abdominal air-sacs ventilated by an expansive sternum and specially hinged costal ribs. The thin walled and voluminous abdominal air-sacs are supported laterally and caudally to prevent inward (paradoxical) collapse during generation of negative (inhalatory) pressure: the synsacrum, posteriorly directed, laterally open pubes and specialized femoral-thigh complex provide requisite support and largely prevent inhalatory collapse. In comparison, theropod dinosaurs probably lacked similarly enlarged abdominal air-sacs, and skeleto-muscular modifications consistent with their ventilation. In the absence of enlarged, functional abdominal air-sacs, theropods were unlikely to have possessed a specialized bird-like, air-sac lung. The likely absence of bird-like pulmonary function in theropods is inconsistent with suggestions of cardiovascular anatomy more sophisticated than that of modern crocodilians.
Science Daily further explains,
It’s been known for decades that the femur, or thigh bone in birds is largely fixed and makes birds into “knee runners,” unlike virtually all other land animals, the [Oregon State University] experts say. What was just discovered, however, is that it’s this fixed position of bird bones and musculature that keeps their air-sac lung from collapsing when the bird inhales.
Warm-blooded birds need about 20 times more oxygen than cold-blooded reptiles, and have evolved a unique lung structure that allows for a high rate of gas exchange and high activity level. Their unusual thigh complex is what helps support the lung and prevent its collapse.
“This is fundamental to bird physiology,” said Devon Quick, an OSU instructor of zoology who completed this work as part of her doctoral studies. “It’s really strange that no one realized this before. The position of the thigh bone and muscles in birds is critical to their lung function, which in turn is what gives them enough lung capacity for flight.”
However, every other animal that has walked on land, the scientists said, has a moveable thigh bone that is involved in their motion — including humans, elephants, dogs, lizards and — in the ancient past — dinosaurs.
The implication, the researchers said, is that birds almost certainly did not descend from theropod dinosaurs, such as tyrannosaurus or allosaurus. The findings add to a growing body of evidence in the past two decades that challenge some of the most widely held beliefs about animal evolution.
“For one thing, birds are found earlier in the fossil record than the dinosaurs they are supposed to have descended from,” Ruben said.
“That’s a pretty serious problem, and there are other inconsistencies with the bird-from-dinosaur theories.
“But one of the primary reasons many scientists kept pointing to birds as having descended from dinosaurs was similarities in their lungs,” Ruben said. “However, theropod dinosaurs had a moving femur and therefore could not have had a lung that worked like that in birds. Their abdominal air sac, if they had one, would have collapsed. That undercuts a critical piece of supporting evidence for the dinosaur-bird link.
Another problem with the claim that birds descended from theropod dinosaurs is that the forelimbs of birds must be able to support their wings and are thus well developed. But the theropods possessed very small forelimbs, which are hardly credible precursors to those of birds and, indeed, a creature like Archaeopteryx. A further difficulty pertains to explaining the origin of feathers. If, as is conventionally maintained, feathers evolved from scales, one has to posit some kind of Darwinian explanation for their evolution from perhaps frayed scales, which must include some kind of genetic basis for every step from frayed scale to fully sculptured feather complete with its many specialized features. Based on what we presently know of the genetic mechanisms, structure and molecular composition of functioning feathers, the proposition that these structures evolved from frayed scales may now reasonably be regarded as highly implausible.
Finally, anyone who has carefully followed the scientific discussion on evolution and the fossil record will doubtless be aware that Archaeopteryx has relatively recently been knocked off its perch, as it were, as the first bird, following the discovery of Xiaotingia zhengi in western Liaoning, China, in rocks dating to the late Jurassic epoch, 161-145 million years ago (Xu et al., 2011). As one news report in Nature put it, “we are about to enter a new era in which Archaeopteryx is considered as distant from the ancestry of modern birds as dinosaurs such as Deinonychus.”
Back to the Water: The Evolution of Whales
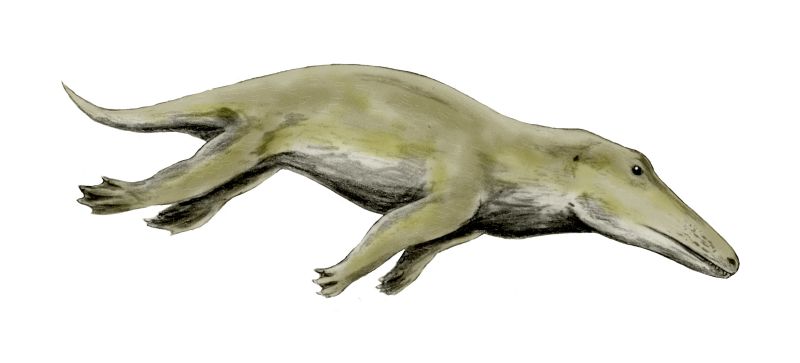 The next transitional series alluded to by Coyne is the whale series. One of the most notable problems with the evolution of the whale is the extremely abrupt timescale over which it is supposed to have occurred. The sheer force of this conundrum is only properly appreciated when one considers the multiple feats of anatomical novelty, innovative engineering and genetic rewiring necessary to change a terrestrial mammal like Pakicetus into a fully aquatic whale. Indeed, evolutionary biologist Richard Sternberg has argued that even many of the relatively minor changes are extremely unlikely to have occurred in the time-frame allowed. Consider the following small sample of necessary modifications:
The next transitional series alluded to by Coyne is the whale series. One of the most notable problems with the evolution of the whale is the extremely abrupt timescale over which it is supposed to have occurred. The sheer force of this conundrum is only properly appreciated when one considers the multiple feats of anatomical novelty, innovative engineering and genetic rewiring necessary to change a terrestrial mammal like Pakicetus into a fully aquatic whale. Indeed, evolutionary biologist Richard Sternberg has argued that even many of the relatively minor changes are extremely unlikely to have occurred in the time-frame allowed. Consider the following small sample of necessary modifications:
- Counter-current heat exchanger for intra-abdominal testes
- Ball vertebra
- Tail flukes and musculature
- Blubber for temperature insulation
- Ability to drink sea water (reorganization of kidney tissues)
- Fetus in breech position (for labor underwater)
- Nurse young underwater (modified mammae)
- Forelimbs transformed into flippers
- Reduction of hindlimbs
- Reduction/loss of pelvis and sacral vertebrae
- Reorganization of the musculature for the reproductive organs
- Hydrodynamic properties of the skin
- Special lung surfactants
- Novel muscle systems for the blowhole
- Modification of the teeth
- Modification of the eye for underwater vision
- Emergence and expansion of the mandibular fat pad with complex lipid distribution
- Reorganization of skull bones and musculature
- Modification of the ear bones
- Decoupling of esophagus and trachea
- Synthesis and metabolism of isovaleric acid (toxic to terrestrial mammals)
- Emergence of blowhole musculature and their neurological control
According to Richard Sternberg’s calculations, and based on the equations of population genetics applied in a 2008 paper by Durrett and Schmidt in the Journal of Genetics, one may reasonably expect to see two coordinated mutations achieve fixation in the timeframe of around 43.3 million years. When one considers the magnitude of the engineering feat, such a scenario can only be ruled incredible. The problem is accentuated further when one considers that the majority of anatomical novelties unique to aquatic cetaceans (Pelagiceti) appeared during just a few million years — probably within 1-3 million years.
Until recently, the whale series, going from Pakicetus (53 mya) to Dorudon and Basilosaurus (38 mya) has looked something like this:
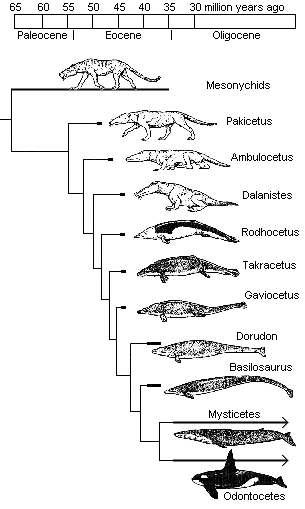
More recently, however, a jawbone was discovered that belonged to a fully aquatic whale dating to 49 million years ago, only four million years after Pakicetus! This means that the first fully aquatic whales now date to around the time when walking whales (Ambulocetus) first appear. This substantially reduces the time window — to 4 or 5 million years, perhaps even less — that may be allotted to the Darwinian mechanism to accomplish truly radical engineering innovations and genetic rewiring. It also suggests that this fully aquatic whale existed before its previously presumed semi-aquatic archaeocetid ancestors.
Lessons from the Fossil Record
According to Coyne, there are three lessons to be learned from the fossil record. The first one is that,
[The fossil record] speaks loudly and eloquently of evolution. The record in the rocks confirms several predictions of evolutionary theory: gradual change within lineages, splitting of lineages, and the existence of transitional forms between very different kinds of organisms. (p. 53)
But the fossil record also exhibits patterns that are not expected under common descent — such as the pattern of saltations followed by stasis, and the appearance of the higher metazoan taxonomic categories before the lower ones.
The second lesson from the fossil record, according to Coyne, is that,
[W]hen we find transitional forms, they occur in the fossil record precisely where they should. The earliest birds appear after dinosaurs but before modern birds. We see ancestral birds spanning the gap between their own landlubber ancestors and fully modern whales. (p. 53)
But this isn’t always the case. In fact, “out-of-sequence” fossils are found routinely. We have already discussed the discovery of tetrapod tracks 20 million years prior to the presumed intermediate fossil linking fish and amphibians. Likewise, Archaeopteryx cannot have descended from the so-called feathered theropod dinosaurs (which exhibit the most bird-like characteristics) because they lived some tens of million years after Archaeopteryx. Actually, these feathered dinosaurs emerge even later than various groups of Cretaceous birds which were more similar to modern ones than was Archaeopteryx. The prevalence of out-of-sequence fossils in the geologic record has led to the regular invocation of “ghost lineages” — see Paul Nelson’s posts here and here for further discussion.
Coyne’s third take-home point from the fossil record is that,
[E]volutionary change, even of a major sort, nearly always involves remodeling the old into the new. The legs of land animals are variations on stout limbs of ancestral fish. The tiny middle ear bones of mammals are remodeled jawbones of their reptilian ancestors. The wings of birds were fashioned from the legs of dinosaurs. And whales are stretched-out land animals whose forelimbs have become paddles and whose nostrils have moved atop their head. (p. 53)
Such remodeling is also seen routinely in human technology. In any case, there are also many instances of convergent evolution, where anatomical and molecular similarities are not readily explicable by common ancestry since the traits emerged some time after the supposed split of the pertinent lineages.
I will review Coyne’s Chapter 3 in a subsequent article.
