 Evolution
Evolution
 Intelligent Design
Intelligent Design
 Medicine
Medicine
How the Body Solves the Problem of Oxygen Transport
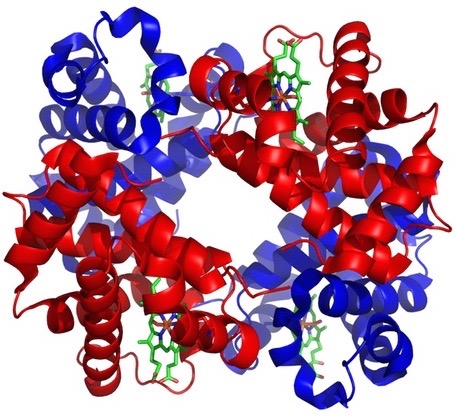
Editor’s note: Physicians have a special place among the thinkers who have elaborated the argument for intelligent design. Perhaps that’s because, more than evolutionary biologists, they are familiar with the challenges of maintaining a functioning complex system, the human body. With that in mind, Evolution News & Views is delighted to present this series, “The Designed Body.” For the complete series, see here. Dr. Glicksman practices palliative medicine for a hospice organization.
 Since all life is built from matter it must follow the laws of nature. These laws demand that to do what it needs to do to survive, life must have enough energy. Our body thus needs, among other things, a constant supply of oxygen (O2). Our lungs breathe in air and pass O2 into the blood. However, that only solves part of the problem. For the laws of nature state that O2 does not dissolve well in water — in fact, only three milliliters (mL) of O2 per liter of water.
Since all life is built from matter it must follow the laws of nature. These laws demand that to do what it needs to do to survive, life must have enough energy. Our body thus needs, among other things, a constant supply of oxygen (O2). Our lungs breathe in air and pass O2 into the blood. However, that only solves part of the problem. For the laws of nature state that O2 does not dissolve well in water — in fact, only three milliliters (mL) of O2 per liter of water.
At rest, the body needs 250 mL/min of O2 and our ancient ancestors would have needed at least 3,500 mL/min of O2 to do what they needed to do to survive. At rest the heart pumps about 5 Liters/minute (L/min) of blood through the body, and with high levels of activity about 25 L/min. If the body were to depend only on its diffusion capacity in blood, there would only be 15 mL/min (3 x 5) of O2 sent to the tissues at rest and only 75 mL/min (3 x 25) with high levels of activity. This represents only 6 percent of what’s needed at rest and only 2 percent of what would have been needed by our ancient ancestors to maintain a level of activity to prey upon others and avoid being preyed upon themselves.
This clearly would have been inadequate. Therefore the body must have developed an innovation. It needed some means to transport O2 more efficiently in the blood.
The body’s solution to the problem of O2 transport is a molecule called hemoglobin. Hemoglobin is produced in developing red blood cells and can carry 1.34 mL of O2 per gram (gm). The blood of a normal adult male has about 150 gm of hemoglobin per liter. Thus his blood can carry about 200 mL of O2 per liter (1.34 x 150) because of the hemoglobin within it.
At rest, since the heart pumps 5 L/min of blood, this means that with the help of hemoglobin, the cardiovascular system can carry 1,000 mL/min of O2 (200 x 5) to the tissues. And with maximum activity, the heart pumps about 25 L/min of blood, and therefore, with the help of hemoglobin, about 5,000 mL/min of O2 (200 x 25) can be transported to the tissues.
It’s important to note that the tissues don’t take all of the O2 out of the blood. They extract about 25 percent to 70 percent. So at rest, 25 percent of the 1,000 mL/min of available O2 will provide the 250 mL/min that the body needs. And with maximum activity, 70 percent of the 5,000 mL/min of available O2 would have provided the 3,500 mL/min our ancestors would have needed to win the battle for survival.
When a person’s level of hemoglobin drops below the normal range, the result is anemia. Worsening anemia causes increasing fatigue, weakness, and shortness of breath with activity. This takes place because, although the lungs may be working properly, the tissues can’t get enough O2 to meet their energy needs due to the blood not being able to carry enough of it.
Clinical experience shows that when the level of hemoglobin drops below the normal range this prevents maximum levels of activity which for our ancestors would have been the difference between eating and being eaten. In addition, since we know that the body requires 250 mL/min of O2 at rest, it is possible to calculate the minimum level of hemoglobin that would generally be compatible with life.
Since the maximum percentage of O2 the tissues can take out of the blood is about 70 percent, there would have to be about 360 mL/min of O2 available (250/0.7). And since, at rest, the heart usually pumps about 5 liters/min, the blood would have to carry 72 mL of O2 per liter (360/5) so that there would be 360 ml/min of O2 available. Finally, since each gram of hemoglobin can carry 1.34 gm of O2, there would have to be a minimum of about 53 grams of hemoglobin per liter of blood (72/1.34) for the body to have the O2 it needs at rest. Thus, all things being equal, a level of hemoglobin that is below 50 gm/L is usually incompatible with life.
Yet at the same time, the body must also be sure not to make too much hemoglobin. After all, hemoglobin is made by the red blood cells that float in the blood. Therefore the laws of nature that govern the flow of liquids must be kept in mind.
Just as having too many food particles in the kitchen sink can clog the drain, so too, having too many red blood cells in the circulation can slow the flow of blood. This slowing of blood flow can clog small blood vessels and, depending on the organs involved, can lead to significant debility and even death.
In other words, not just any number of red blood cells making just any amount of hemoglobin is sufficient for life. It’s important for the body to maintain control of how many red blood cells and how much hemoglobin it makes. How does the body do this?
As we’ve seen before in this series, the first thing needed to take control is a sensor that can detect what has to be controlled. There are specialized cells within the kidneys that are thought to detect the O2 level within the blood, which is directly related to its content of hemoglobin.
The second thing needed to take control is an integrator that analyzes the information from the sensors and sends out orders. The same cells in the kidneys that detect O2 also send out a hormone called erythropoietin based on the data they receive from the sensors. The third thing needed to take control is an effector that receives the orders from the integrator and does something.
When erythropoietin locks onto the specific receptors on the immature cells in the bone marrow, this signals them to mature into red blood cells and start making hemoglobin. How the specialized cells in the kidneys know how much erythropoietin they should release for a given O2 level, and how the red blood cells know how much hemoglobin they should make so the body can transport enough O2 to the tissues, is as yet a complete mystery.
Now, considering all of the parts and the controls needed just to get enough O2 to all of your cells, do you think that this could have all come about by chance and the laws of nature alone? Indeed, how can evolutionary biologists explain the development of these control systems that seem to inherently know what is needed for us to survive, if medical science doesn’t even understand them yet?
However, there’s more. Having the right amount of hemoglobin requires the presence of a metal called iron. Not having enough iron can result in what is called iron deficiency anemia. But having too much iron can be toxic to the body as well. Next week we’ll look at how the body acquires iron and, by following the rules, takes control of it so that real numbers won’t result in death.
Image: Human hemoglobin, by Zephyris at the English language Wikipedia [GFDL or CC-BY-SA-3.0], via Wikimedia Commons.
