 Evolution
Evolution
 Intelligent Design
Intelligent Design
A Simple Transition to Multicellularity — Not!

Volvox is a small spheroid green alga that lives in ponds, making its living by photosynthesis as plants do. Volvox is among the simplest animals to have more than one cell type. The hollow sphere that is its body is made up of somatic cells (soma is the Greek word for body). Within the sphere are specialized cells called gonidia (from the Greek gon?, meaning generation or seed). Early in embryogenesis, some cells divide to produce large cells. These become the gonidia, which then become embryos with their own seeds of gonidia. Eventually the embryos mature into the next generation of Volvox, with their own embryos growing inside.
Interestingly, though, when times get hard and the pond starts to dry up or things get too hot, the embryos develop into female and male forms that produce eggs and sperm rather than divide asexually. These eggs and sperm fuse to make new diploid Volvox, which then goes into a sort of stasis until the water returns. That’s a neat trick. Then the asexual cycle starts all over again.
Volvox somatic cells each have two flagella. The whip-like flagella help these little spheroid creatures to swim in a sort of gentle ballet. See here:
The somatic cells are embedded in an extracellular matrix, a gel-like substance that supports and surrounds the cells. The somatic cells in the video appear as an outer green haze on the spinning spheres. The bright green small spheres within the bodies of the Volvox are the developing embryos.
Volvox is often used as an example of how the evolution to multicellularity might have happened. The story is that single flagellate cells like C. reinhardtii (which I wrote about here earlier this week) first bundled together, then the bundled cells took on a more organized shape, perhaps to aid swimming, then those groups developed several cell types that could divide the labor among themselves. The beginning of more complex life forms, perhaps?
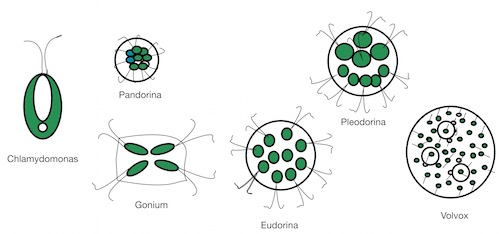
To hear the story told, it seems a simple progression. Only one or two or genes might be necessary to get the cells to form clumps by incomplete cell divisions. There are species that look like this stage — clumps of cells that swim and then stop swimming and divide. How those cells separate is another question. A few more hypothetical genes would be necessary to make the extracellular matrix in which most species embed their cells and then to digest it when the right time comes. Then the final essential step is taken, the division of labor between somatic and reproductive cells. Some cells grow into little embryos and then adults, while other cells adopt a supportive role, contributing food to the growing embryos inside. A theory has been proposed involving 12 steps for how this change from single-celled to multicellular Volvox might have happened.
Missing from this story, though, are the details necessary for this 12-step progression to occur. There’s the matter of incomplete cell division, cell separation later, and matrix formation. Then there’s the matter of specialization. To get the specialization of somatic from germ cells requires the development of at least three proteins. Also, the embryos produced by those reproductive cells are inside out — the flagella are on the inside, which makes them useless for swimming, and the gonidia are on the outside. As a result, the embryo must turn itself inside out when it reaches the right size. This requires that the somatic cells change shape to bottle- or spindle-shaped cells, depending on their position in the embryo. This shape change requires both microtubules and a little motor protein called kinesin. Neither microtubules or kinesin are present in bacteria so their origin must be accounted for. The selective advantage of each step is not clear.
Later, the parent Volvox produces an enzyme that digests the matrix the somatic cells are embedded in, and releases the embryos. The somatic cells commit suicide by a process known as apoptosis — programmed cell death — that I wrote about here. This process involves a minimum of several novel genes as well.
Finally, there is the not so small matter of sexual reproduction. Getting two kinds of reproductive cells, eggs and sperm, requires triggering a new set of regulatory genes to change the pattern of gene expression, so as to produce the two cell types. And more than that — going into stasis, recovering from stasis, and going from diploid to haploid again requires yet more regulation. (Volvox normally has only one set of chromosomes — they are haploid — but after fusion of egg and sperm they are diploid — having two sets of chromosomes.)
Does the story seem simple now? When you think of it as just incomplete divisions and a three-gene regulatory scheme coopted from other jobs to cause the switch from somatic to reproductive cells, it sounds plausible. When you add the other requirements, though, the story becomes untenable. A coordinated process like inversion requires specific cell shape changes signaled by some process, and requiring microtubules and kinesin at a minimum. (Both of these proteins are necessary for multiple other cellular processes, by the way, but are new to eukaryotes.) And remember, inversion is a necessary process — without it the embryos can’t swim. Next the digestive enzyme must be produced at the right time, which no doubt involves some signaling process, and all the (new) proteins involved in programmed cell death (also new to eukaryotes) must be made and/or activated.
Where do all these new proteins come from? Either they come from cooption of old proteins, or by making new ones. I’ve already been over how hard those processes are to accomplish multiple times. Where does their regulation come from? That involves yet more proteins to serve as genetic switches, which also must be coopted or made from spare sequence lying around. It’s an infinite regress of steps to be filled, and answering one question about mechanism leads to many more.
I think asking these questions is well worth the effort. With each answer found we learn something more about the way things work in living things, and we find similarities and differences and discern underlying principles. But what I object to is the evolutionary gloss put on these similarities and differences, these principles. It’s one thing to say things were coopted from some unknown precursor protein, and another to demonstrate that such cooption is possible.
This is the sort of detail that requires answering if the white space in evolutionary thinking is to be filled. Saying that something might have happened is not the same as showing that it actually could happen.
Image: Volvox, by Frank Fox (http://www.mikro-foto.de) [CC BY-SA 3.0 de], via Wikimedia Commons.
