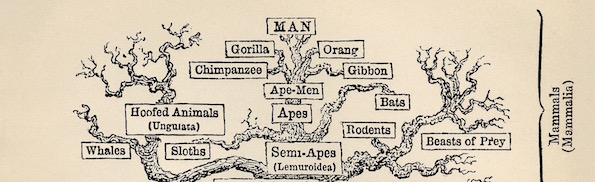
Evolution
Evolution
 Intelligent Design
Intelligent Design
Do All Life Forms Fall into a Nested Hierarchy?
A biology graduate student and alumnus of our Summer Seminar on Intelligent Design recently contacted me to ask where ID comes down on common descent. I explained to him that in the ID camp, some folks accept common descent while others are skeptical. ID proponents can respectfully disagree on that question (and others too), while agreeing on the powerful evidence for design in nature. But my correspondent asked about an argument in favor of common ancestry he had heard that basically went like this:
All life forms fall within a nested hierarchy. Of the hundreds of thousands of specimens that have been tested, every single one falls within a nested hierarchy, or their evolutionary phylogenetic tree is still unknown and not sequenced yet.
This claim (which he wasn’t making, by the way) is far from true. We constantly find organisms that don’t fit neatly into a phylogenetic tree. Or, what happens is evolutionary biologists attempt to force-fit organisms into the tree only by invoking processes like convergent evolution and loss of traits. In other words, evolutionary biologists are forced to propose that an organism’s traits did not arise through common ancestry, because common ancestry fails to explain the data.
Does this mean that evolutionary biologists reject common ancestry when they find data that doesn’t fit a tree? No, because they assume common ancestry; they aren’t interested in testing it. So when they find data that doesn’t fit a tree, they just find ways to force-fit the data into the tree. Here’s what’s going on:
The first and primary assumption of all evolutionary phylogenetic classification methodologies is that common ancestry is true. This assumption nearly always goes unquestioned, even when the data doesn’t support it. As Elliott Sober and Michael Steele explain, “It is a central tenet of modern evolutionary theory that all living things now on earth trace back to a single common ancestor,” and “This proposition is central because it is presupposed so widely in evolutionary research.” They acknowledge that cladistics assumes that a tree exists, and common ancestry is correct:
Whether one uses cladistic parsimony, distance measures, or maximum likelihood methods, the typical question is which tree is the best one, not whether there is a tree in the first place.
(Elliott Sober and Michael Steele, “Testing the Hypothesis of Common Ancestry,” Journal of Theoretical Biology 218 (2002): 395-408 (emphasis added).)
Likewise, the assumption is made explicit, and primary, in the UC Berkeley Museum of Paleontology’s introductory page on cladistics:
What assumptions do cladists make?
There are three basic assumptions in cladistics:
1.Any group of organisms are related by descent from a common ancestor.
One textbook cited by Stephen Meyer in Darwin’s Doubt concurs about this assumption:
The key assumption made when constructing a phylogenetic tree from a set of sequences is that they are all derived from a single ancestral sequence, i.e., they are homologous.
(Marketa Zvelebil and Jeremy O. Baum, Understanding Bioinformatics (New York: Garland Science, 2008), p. 239.)
Together, these authorities make a crucial point: cladistics and other phylogenetics methods do not demonstrate common ancestry; they assume it. In other words, these methods don’t test whether all organisms fit into a nested hierarchy (i.e., phylogenetic tree). Rather, evolutionary systematics assumes that common ancestry is true and therefore all organisms belong within a nested hierarchy, and then it uses methods to force-fit any organism into the tree, even if that organisms has traits that don’t fit neatly within the tree. Thus, Michael Syvanen — a rare evolutionary biologist who is open to the possibility that universal common ancestry is false — laments the pro-tree biases of treebuilding algorithms:
Because tree analysis tools are used so widely, they tend to introduce a bias into the interpretation of results. Hence, one needs to be continually reminded that submitting multiple sequences (DNA, protein, or other character states) to phylogenetic analysis produces trees because that is the nature of the algorithms used.
(Michael Syvanen, “Evolutionary Implications of Horizontal Gene Transfer,” Annual Review of Genetics, 46:339-356 (2012) (emphases added).)
Common ancestry, therefore, is a starting assumption about the data — not a conclusion from it. Another key lesson is this: just because you see evolutionary biologists creating an impressive-looking phylogenetic tree doesn’t mean that all of the organisms or their traits shown within that tree fit neatly into a nested hierarchy (i.e., a tree structure). One could cite many examples of organisms that don’t fit cleanly into a tree. Here are a few:
Sahelanthropus tchadensis is widely touted as a human ancestor that lived about 6-7 million years ago, sometime very soon after the supposed split between the human line and the chimp line. But it’s rarely mentioned that this specimen doesn’t fit into the standard hominin tree at all. Why? Because it has a flat face, a humanlike quality, which shouldn’t exist that far back:
If we accept these as sufficient evidence to classify S. tchadensis as a hominid at the base, or stem, of the modern human clade, then it plays havoc with the tidy model of human origins. Quite simply, a hominid of this age should only just be beginning to show signs of being a hominid. It certainly should not have the face of a hominid less than one-third of its geological age. Also, if it is accepted as a stem hominid, under the tidy model the principle of parsimony dictates that all creatures with more primitive faces (and that is a very long list) would, perforce, have to be excluded from the ancestry of modern humans.”
(Bernard Wood, “Hominid revelations from Chad,” Nature, 418 (July 11, 2002):133-35.)
Because of this, some are skeptical that S. tchadensis belongs on the human line. If that’s the case then its flat face represents convergent evolution. And if it is on the human line, then you are forced to propose that later species on the human line lost this trait. Either way, S. tchadensis creates major problems for a nice, neat, nested hierarchy of hominins that is consistent with the chronology of the fossil record.
Much further back, there are organisms like Diania, thought to be an early arthropod ancestor from the Cambrian period, but which actually cause huge problems for the arthropod tree:
[W]e should caution that dinocaridids, Diania and other potential stem-arthropods typically express mosaics of arthropod-like characters, which makes resolving a single, simple tree of arthropod origins problematic. Indeed, the position recovered here for Diania between Radiodonta and the ostensibly similar-looking Schinderhannes is surprising. Diania, Schinderhannes and the remaining Arthropoda all share the putative apomorphy of jointed trunk appendages, and yet the trunk limbs of Diania resemble the frontal appendages of Anomalocaris and other radiodontans, which themselves lack trunk limbs entirely. If this is a secondary reduction in fossils like Anomalocaris, then Diania may in fact occupy a more basal position with respect to Radiodonta, Schinderhannes and Arthropoda; a scenario that would be more consistent with their fairly simple body morphology.
(Liu et al., “An armoured Cambrian lobopodian from China with arthropod-like appendages,” Nature, 470 (February 24, 2011): 526-530 (internal citations omitted).)
The reason the authors talk about “mosaics of arthropod-like characters” is that these organisms don’t fit into an orderly, sequential, hierarchical, treelike pattern as predicted by common descent. They present a mishmash of traits, not distributed in a treelike pattern that shows some sequential, hierarchy ordering arthropod traits. This is a famous problem in arthropod evolution, rightly described as a mess. As one paper observes: “Arthropod phylogeny is sometimes presented as an almost hopeless puzzle wherein all possible competing hypotheses have support.” That’s the opposite of a nested hierarchy.
Nor is it just within the Cambrian phyla that we find such an array of phylogenetic misfits. Among the animal phyla more generally, we see traits that make it difficult to create a treelike representation of relationships.
The argument cited by the grad student noted that one reason an organism’s classification isn’t understood is that “their evolutionary phylogenetic tree is still unknown and not sequenced yet.” That’s a bit of a rudimentary argument, but it probably means that until we sequence an organism’s genome, we don’t know where it belongs in the tree of life. But sometimes after we sequence an organism’s genome we find that its place in the tree of life is even less clear than it was before.
This is exactly what happened after the comb jelly genome was sequenced. As I wrote last year, comb jellies (phylum ctenophora) have muscles and complex nervous systems, but molecular studies suggest they branch off very close to the base of the animal tree. However, sponges — which branch off later according to molecular data — lack such structures. This means that either complex muscle and nervous cells were lost in sponges (even though these are complex, useful traits you’d probably want to keep around) or muscles and brains evolved convergently in later animals. Either way, you’re left with a situation where comb jellies don’t fit neatly into the animal tree. They show a mosaic of traits that shouldn’t be the case under common descent.
Here’s one more classic example from the animal phyla: symmetry. Animals can be divided up in many different ways. Some display bilateral symmetry, basically meaning they have a right half and a left half. Such “bilaterian” phyla include vertebrates, arthropods, and mollusks. Others have radial symmetry, where their symmetry is distributed in an essentially circular fashion around a central axis. Phyla with radial symmetry include cnidarians (e.g., jellyfish), ctenophores (comb jellies), and echinoderms (e.g., star fish and sea urchins).
From an evolutionary perspective, you might expect an animal tree to divide up neatly according to whether organisms display bilateral or radial symmetry. Not so. Echinoderms are placed much closer to vertebrates than they are to cnidarians and other phyla with radial symmetry. In fact, in another weird twist, vertebrates (with bilateral symmetry) are thought to be much closer to echinoderms (with radial symmetry) than they are to other bilaterian phyla like the arthropods, mollusks, or annelids. This grouping is made on the basis of early developmental processes. Echinoderms and vertebrates are both deuterostomes, meaning that early in development the first opening in the blastopore becomes the anus rather than the mouth. The upshot is this: animal symmetry is not distributed in a treelike pattern.
Yes, a critic might object that larval stages of echinoderms can have bilateral symmetry. But exactly the same can be said of some cnidarians, which are very far from echinoderms in the animal tree — showing, again, that symmetry is not distributed in a treelike pattern.
In any case, an evolutionary biologist could decide to group phyla according to early developmental processes, or according to symmetry, and that’s fine. If you weight one trait heavily, you’ll get one tree. But switch that weight to another trait and you’ll get another, conflicting tree. Either way, when you use one character set to create your tree, then the other character set is no longer distributed in a treelike fashion, and vice versa. That’s a major problem.
Another good example of an organism whose genome posed problems for phylogenetic classification after it was sequenced is birds. As we reported last December, the sequencing of various bird genomes led to the unexpected conclusion that many types of birds that were previously thought to be closely related — water birds, birds of prey, and songbirds — evolved their groups’ defining traits convergently. As Nature put it, “the tree of life for birds has been redrawn” by this study. The problem was, once genomic data was sequenced and understood, many basic habits and lifestyles of birds no longer fit into a nested hierarchy.
Again, there are innumerable examples of organismal traits that don’t fit into a treelike pattern. But here are two more that came out recently.
In April, Science Daily reported a study that looked at how different marine organisms swim (“Convergent evolution: Diverse sea creatures evolved to reach same swimming solution“). The scientists found that organisms as diverse as cuttlefish (a mollusk), the black ghost knifefish (a vertebrate), and the Persian carpet flatworm (phylum platyhelminthes) all use the same method of swimming:
The ability to move one’s body rapidly through water is a key to existence for many species on this blue planet of ours. The Persian carpet flatworm, the cuttlefish and the black ghost knifefish look nothing like each other — their last common ancestor lived 550 million years ago, before the Cambrian period — but a new study uses a combination of computer simulations, a robotic fish and video footage of real fish to show that all three aquatic creatures have evolved to swim with elongated fins using the same mechanical motion that optimizes their speed, helping to ensure their survival.
These three animals are part of a very diverse group of aquatic animals — both vertebrate and invertebrate — that independently arrived at the same solution of how to use their fins to maximize speed. And, remarkably, this so-called “convergent” evolution happened at least eight times across three different phyla, or animal groups, supporting the belief that necessity played a larger role than chance in developing this trait.
Now they are welcome to invoke convergent evolution if they like, but it’s striking how each of these widely diverse organisms has a very similar mode of swimming, where the length of one undulation of the animal’s fins divided by the average amplitude of the corresponding sideways movement gives you a ratio of about 20. You can see how distantly related (according to the usual evolutionary paradigm) these similar-swimming organisms are by looking at a tree diagram from the original paper.
Also in April there were striking reports of a new vegetarian theropod dinosaur that had traits that made it very difficult to classify within the standard dinosaur tree:
Although closely related to the notorious carnivore Tyrannosaurus rex, a new lineage of dinosaur discovered in Chile is proving to be an evolutionary jigsaw puzzle, as it preferred to graze upon plants. Chilesaurus boasted a proportionally small skull, hands with two fingers like Tyrannosaurus rex and feet more akin to primitive long-neck dinosaurs.
Theropods, of course, include the meat eaters Velociraptor and T. rex, yet this species was a vegetarian. It thus poses a severe puzzle for evolutionary classification, as an article in The Guardian acknowledged:
Fossil hunters in Chile have unearthed the remains of a bizarre Jurassic dinosaur that combined a curious mixture of features from different prehistoric animals.
The evolutionary muddle of a beast grew to the size of a small horse and was the most abundant animal to be found 145 million years ago, in what is now the Aysén region of Patagonia. The discovery ranks as one of the most remarkable dinosaur finds of the past 20 years, and promises to cause plenty of headaches for paleontologists hoping to place the animal in the dinosaur family tree.??”I don’t know how the evolution of dinosaurs produced this kind of animal, what kind of ecological pressures must have been at work,” said Fernando Novas at the Bernardino Rivadavia Natural Sciences Museum in Buenos Aires.
The technical paper in Nature puts it this way:
For a basal tetanuran, Chilesaurus possesses a number of surprisingly plesiomorphic traits on the hindlimbs, especially in the ankle and foot, which resemble basal sauropodomorphs. These features are here considered as secondary reversals that might be related to a less-cursorial mode of locomotion. Furthermore, derived features of the dentary and teeth shared by Chilesaurus, sauropodomorphs and therizinosaurs are interpreted as homoplasies related to herbivorous habits. …
Chilesaurus represents an extreme case of mosaic evolution among dinosaurs, owing to the presence of dental, cranial and postcranial features that are homoplastic with multiple disparate groups. Using quantitative morphospace analysis, we explored morphospace occupation of different skeletal regions in Chilesaurus with respect to a variety of avian and non-avian theropods. This shows that Chilesaurus has a ceratosaur-like axial skeleton, a ‘basal tetanuran’ forelimb and scapular girdle, a coelurosaur-like pelvis, and a tetanuran-like hindlimb. General ankle and foot construction does not group with any theropod clade, probably as a result of the characters shared by Chilesaurus, sauropodomorphs and herrerasaurids.
Science Daily explained in less technical terms why this species, with its set of traits resembling many different types of dinosaurs, is difficult to classify:
Other features present in very different groups of dinosaurs Chilesaurus adopted were robust forelimbs similar to Jurassic theropods such as Allosaurus, although its hands were provided with two blunt fingers, unlike the sharp claws of fellow theropod Velociraptor. Chilesaurus‘ pelvic girdle resembles that of the ornithischian dinosaurs, whereas it is actually classified in the other basic dinosaur division — Saurischia.
The different parts of the body of Chilesaurus were adapted to a particular diet and way of life, which was similar to other groups of dinosaurs. As a result of these similar habits, different regions of the body of Chilesaurus evolved resembling those present in other, unrelated groups of dinosaurs, which is a phenomenon called evolutionary convergence.
Chilesaurus represents one of the most extreme cases of mosaic convergent evolution recorded in the history of life. For example, the teeth of Chilesaurus are very similar to those of primitive long-neck dinosaurs because they were selected over millions of years as a result of a similar diet between these two lineages of dinosaurs.
Martín Ezcurra, Researcher, School of Geography, Earth and Environmental Sciences, University of Birmingham said: ‘Chilesaurus can be considered a ‘platypus’ dinosaur because different parts of its body resemble those of other dinosaur groups due to mosaic convergent evolution. In this process, a region or regions of an organism resemble others of unrelated species because of a similar mode of life and evolutionary pressures. Chilesaurus provides a good example of how evolution works in deep time and it is one of the most interesting cases of convergent evolution documented in the history of life.
What we see here is a dino that doesn’t fit with the dino tree not just because it’s a herbivorous theropod, but also because different parts of its body appear similar to different types of dinosaurs. Through convergent evolution and loss of traits, you can always find a way accommodate such quirky data. However the bottom line is that organisms like these are the opposite of finding “All life forms fall within nested hierarchy.”
We explain further in our curriculum Discovering Intelligent Design:
You may recall the “main assumption,” mentioned at the beginning of the chapter, that Darwinian evolutionists use when constructing trees: similarity implies inheritance from a common ancestor. But what about situations where that assumption is clearly untrue — i.e., when two organisms share a trait that their supposed common ancestor could not have possessed?
A striking example is the [skull] similarity between marsupial and placental “saber-toothed cats,” which are classified as very different types of mammals due to their two distinctly different ways of bearing young.
According to current evolutionary theory, the common ancestor of these two cats was a small rodent-like mammal with a very different body plan. Thus, their highly similar skull structures had to develop independently and could not have been inherited from a common ancestor.
Common descent does not explain these similarities. Evolutionists try to explain this evidence by claiming these distinctly different cats evolved the same traits independently through convergent evolution.
Does this sort of data absolutely refute universal common descent? Taken on a case-by-case basis, no of course not, and we’re not claiming it does. What it shows, collectively, is that the evolutionary case is a lot weaker than is routinely claimed. We commonly — if not constantly — find organisms whose traits don’t fit into a hierarchical tree.
I like how we conclude on this topic in Discovering Intelligent Design:
Perhaps the main assumption of phylogenetic trees should be rewritten as: “similarity implies inheritance from a common ancestor, except when it does not.” Rather than being a helpful solution for neo-Darwinists, convergent evolution undermines the reasoning used to construct phylogenetic trees.
In fact, maintaining that “All life forms fall within a nested hierarchy” requires you to ignore huge amounts of data. ID proponents who doubt common descent have, I would say, ample reason for doing so.
Image: Tree of Life, by Ernst Haeckel [Public domain], via Wikimedia Commons.