 Evolution
Evolution
 Intelligent Design
Intelligent Design
Michael Behe’s Challenge — Past, Present, and Future
Twenty years after Darwin’s Black Box, the irreducibly complex outboard motors showcased by Michael Behe still confound Darwinism. That’s one theme of our upcoming documentary Revolutionary: Michael Behe & The Mystery of Molecular Machines, which heads to Texas next month for previews. Behe’s challenge is the past, the present, and the future.
Look at this quote from a paper in the Proceedings of the National Academy of Sciences:
The bacterial flagellar system has been an object of intense study for many years. It has helped to elucidate issues of assembly, motility, and chemotaxis at a molecular level in a relatively simple system, typically containing ∼40 different proteins. It has also been the icon for creationists in the United States who deny evolution. [Emphasis added.]
Ah, the bacterial flagellar system. The paper by scientists from Germany, the Netherlands, Israel, and the U.S. (University of Virginia) purports to show how a simpler flagellum in a species of archaea provides evidence for evolution of the more complex flagellum in bacteria. It’s satisfying to reflect that the authors remain irritated by the “creationist” icon. They can’t let it rest.
As expected, they refuse to cite Behe directly (or any other ID source), so their only references supporting the “creationist” charge are to 6-to-12-year-old writings by Kenneth Miller, Barbara Forrest, and E.H. Egelmann. Have it your way. What matters is the substance of Behe’s argument and evidence, which attracted the attention of researchers in four nations.
Meanwhile, farther east — particularly in Japan — scientists don’t seem quite so motivated to defend Darwin. Two new papers dealing with molecular motors show their openness to design thinking.
Cilium Mimicry
The first deals with designing a mimic of cilia, another one of Behe’s examples of irreducibly complex (IC) systems. Five biomedical engineers from Tokyo, writing in Science Advances, succeeded in making “Artificial cilia as autonomous nanoactuators.” They call this “Design of a gradient self-oscillating polymer brush with controlled unidirectional motion.”
Here, we have prepared a polymer brush surface similar to a living cilium, exhibiting self-oscillating and unidirectional wave motion of the grafted polymer at the nanometer scale…. This study provides a new concept to design autonomous polymer brush surfaces effective in the nanometer scale as bioinspired dynamic soft materials.
They pay tribute to the design in cilia and other cellular machines that inspired their own work:
Spatiotemporally well-ordered mechanical actuation of biomacromolecules in the nanometer-order scale driven by chemical reactions, such as enzymatic reactions, plays an important role in living organisms. For example, motor proteins bind to a polarized cytoskeletal filaments and use the energy derived from repeated cycles of adenosine 5′-triphosphate hydrolysis to move steadily along them. In addition, many motor proteins carry membrane-enclosed organelles to their appropriate locations in the cell. Cytoskeletal motor proteins move unidirectionally along an oriented polymer track. In this process, they use chemical energy to propel themselves along a linear path, and the direction of sliding is dependent on the structural polarity of the track. Recently, the construction and design of these biomolecular motor systems with well-controlled unidirectional motion have become an area of great focus in advanced sciences.
The word “design” appears ten times in the paper. References to evolution are completely absent.
Another team worked on imitating the cilia in hair cells of the inner ear. Publishing in Nature‘s journal Scientific Reports, they proudly announce, “Here we present a novel, completely biomimetic flow sensor which attempts to replicate the intricate morphological organization and function of the hair bundles of the hair cells” found in zebrafish. Neither of the cheap imitations, however, contain anything like intraflagellar transport in biological cilia (see our report). As for the origin of these intricate structures, the team treated evolution as magic: “Nature’s evolutionary path led to sensors of high functionality and robustness, in terms of material properties, anatomical architecture and energy expenditure.” That’s all the press Darwin got. They used the word design, though, 15 times.
Flagellum Focus
Back in the 1990s, Behe relied on relatively crude electron micrographs of flagella. Imagine twenty years ago if he had been able to see in detail one protein in the stator of the flagellum. That’s what Japanese scientists from Nagoya University revealed with advanced imaging techniques. So has the case for design grown stronger or weaker since Darwin’s Black Box was published? A paper in Nature‘s open-access journal Scientific Reports reveals the answer. You can see their composite image of the stator protein MotA here:
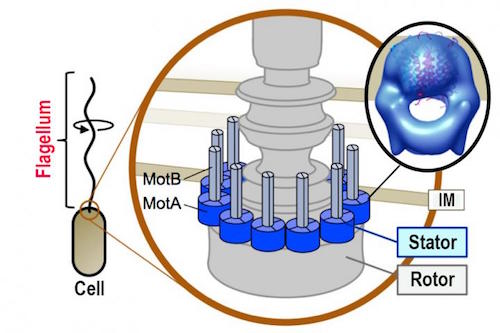
It looks more like a well-designed outboard motor than ever!
Many bacterial species use spiral propellers (flagella) attached to motors to move through a liquid environment. An interaction between the rotor and stator components of the motor generates the rotational force required for movement. The stator converts electrochemical energy into mechanical force after undergoing a structural change caused by a movement of charged particles (ions) through an internal channel. Previous studies investigated the stator and its interaction with the rotor by constructing mutant proteins and analyzing their functions. However, little was known about stator structure.
A team of Japanese researchers led by Homma’s laboratory of Nagoya University have now purified the stator protein MotA from a bacterium found in hot springs (Aquifex aeolicus) and analyzed its three-dimensional structure using electron microscopy mainly in cooperation with Namba’s laboratory of Osaka University.
The stator protein MotA shows an elegantly crafted channel for ions. These are arranged in groups of four at the base of the stator on the cytoplasmic side. Two slender molecules of MotB extend into the periplasm. Identifying the structure is an important step on the way to figuring out how the flagellum works.
The stator is one of the most important parts for the proper functioning of the bacterial flagellar motor, and is believed to work as an energy-converting unit that transduces electrochemical potential gradient across the cytoplasmic membrane into mechanical force. The interaction surfaces of the stator and the rotor have been well studied by mutational analyses. However, the mechanism governing energy conversion remains unknown because of the lack of structural information on the stator.
How is chemical energy converted into rotational energy (torque)? “It is believed that the ion influx through the channel induces a conformational change in the stator, allowing its interaction with the rotor to generate torque,” they say.
A little reflection suggests that the components must be well matched. Imagine for a moment a flow of ping-pong balls trying to turn a large metal turbine. It wouldn’t work. Somehow, in ways still to be determined two decades after Behe’s book was published, tiny hydrogen ions or sodium ions are able to make particular parts of the stator undergo conformational changes that can get the rotor to turn. How that works is bound to be interesting. We know from the discussion in Unlocking the Mystery of Life that not only must the parts be well matched, they must be assembled in the right order, in the right amounts, at the right place and time — all directed by instructions in the genetic code.
While these Japanese researchers do not mention Behe or his conclusions, their work underscores the case for irreducible complex molecular machines as prima facie evidence for intelligent design. Nowhere do they credit evolution for the motors of the cell:
Various motor proteins are essential for different biological activities such as cell locomotion, cell morphogenesis, metabolism, and material transport. Motor proteins convert various types of energy, such as ATP hydrolysis or electrochemical potential, into mechanical force for directional motion. Motor proteins are divided into two types: linear motors, such as myosin-actin and kinesin/dynein microtubule motors; and rotary motors, such as ATP synthase and bacterial flagellar motor.
Behe’s intuitive first impression of the flagellum, “That’s an outboard motor!” has stood the test of time. That’s what the Japanese teams still call the cilium and the flagellum in 2016.
Scratching the Itch
Now back to the PNAS paper. Did the Western nations solve Michael Behe’s challenge? If so, they have a strange way of claiming success:
The proteins that form the bacterial flagellar system have no known homologs in eukaryotic cells. The eukaryotic flagellar [sic], based on a microtubule-containing axoneme, is vastly more complicated. In fact, the current estimate for the number of different proteins in the axoneme is ∼425. In contrast, the archaeal flagellar system appears simpler than the bacterial one and can contain as few as 13 different proteins. As with the eukaryotic flagellar system, the archaeal one does not have homology with the bacterial one and must have arisen by means of convergent evolution.
Ah yes, convergent evolution again. But think about what they say here. The “vastly more complicated” eukaryotic flagellum has no known commonalities with the bacterial flagellum, and the bacterial flagellum has no homolog in the archaeal flagellum: “In archaeal flagellins, however, no homology has yet been found outside of the N-terminal domain with any bacterial or eukaryotic proteins.” Do they show any common ancestry between these motors? None. Are we to believe, then, that blind processes happened upon three naturalistic miracles independently?
Notice that the archaeal flagellar system that “appears simpler” contains as few as 13 different proteins. How many different parts were required for the mousetrap? Behe’s pithy illustration of an IC system had only five parts. How much more is an IC system with 13 parts, or with 40 parts, each unrelated to machines with similar functions in other types of cells?
If the best that evolutionists can come up with in response to Behe is “convergent evolution,” then his 1996 statement can be seen as prescient: “An irreducibly complex biological system, if there is such a thing, would be a powerful challenge to Darwinian evolution” (p. 39). Only he could reword it now with much more confidence: Irreducibly complex biological systems continue to pose a powerful challenge to Darwinian evolution and are best explained by intelligent design.
Image: Composite image of the stator protein MotA, by Michio Homma via EurekAlert.

