Evolution
Evolution
 Faith & Science
Faith & Science
 Intelligent Design
Intelligent Design
On the Cambrian Explosion, Keith Miller’s BioLogos White Paper Falls Short

The Cambrian explosion is a major mystery for Darwinian evolution, so it may be no surprise that the theistic evolutionary group BioLogos has compiled a mixed record in analyzing the event. Back in 2010, we noted that the BioLogos page on the fossil record didn’t even mention the Cambrian explosion. Then, in response to Stephen Meyer’s 2013 book Darwin’s Doubt, reviewers for BioLogos struggled to come to a consensus. Instead they reached conflicting conclusions.
Some claimed that the Cambrian explosion poses no challenge to traditional neo-Darwinian explanations of life’s origins. On the other hand, BioLogos past president Darrel Falk admitted, “The big mystery associated with the Cambrian explosion is the rapid generation of body plans de novo.” Dr. Falk also conceded that Stephen Meyer was correct to say that neo-Darwinism faces serious criticism in the mainstream scientific community.
Overall, however, for dealing with the Cambrian explosion, papering over other deficiencies in evolutionary explanations, and sidestepping evidence for intelligent design, the main strategy at BioLogos has been to invoke methodological naturalism.
Recently, a reader brought to our attention a BioLogos post by Kansas State University geologist Keith Miller, “The Cambrian ‘Explosion,’ Transitional Forms, and the Tree of Life.” The article appeared in 2010 and is intended as an authoritative “white paper” of sorts. It is part one of a series at BioLogos on the Cambrian explosion. See here for a PDF.
Miller’s article seems to be based partly upon an earlier article he wrote for the American Scientific Affiliations which claims there are Precambrian “transitional forms” documenting the evolution of the Cambrian phyla. Though the paper is several years old and, with apologies, we’re only now getting around to writing about it, we have long been aware of Miller’s style of argument. In fact, nearly all of the issues he raises are addressed in Darwin’s Doubt or Debating Darwin’s Doubt. But for the reader who asked about it, what Miller had to say was evidently new, and so it must be for others who are just catching up on the discussion of the Cambrian mystery. We thus take this opportunity to answer his claims.
A Long Fuse
A major part of Miller’s argument is to claim that the Cambrian explosion was a long event that lasted many tens of millions of years (his preferred number is “52 million years”), This, he thinks, rebuts Stephen Meyer’s argument that “the fauna of the Cambrian period appeared in a geologically sudden manner.” We have addressed this kind of argument many times before. We have pointed out the consensus view within the scientific literature that the Cambrian explosion lasted about 10 million years. Numerous authorities have acknowledged the Cambrian explosion was geologically “sudden.” Evolutionary biologists may thus seek to redefine the “Cambrian explosion” by expanding the temporal or taxonomic boundaries. Yet none of that changes the fact that there occurred an intense burst of novelty in the early-middle Cambrian — a burst lasting only about 5 to 10 million years.
Miller cites the small shelly fauna from the early Cambrian, claiming that they indicate a lengthy period of time for the evolution of the Cambrian animals. But as we have discussed, many authorities note that these “largely problematic fossils” are “hard to diagnose, even at the phylum level” and are “are very difficult indeed to interpret.” The enigmatic small shelly fossils don’t clearly map on to other Cambrian animals.
Miller similarly claims the Cambrian explosion had a “long fuse” — extending back into the Precambrian, where we see “transitional fossils” leading to Cambrian animals. But as Stephen Meyer documents in Darwin’s Doubt, “with the exception of sponges and the possible exception of Kimberella, the body plans of visibly fossilized [Precambrian] organisms (as opposed to trace fossils) bear no clear relationship to any of the organisms that appear in the Cambrian explosion (or thereafter).” Even Keith Miller cannot deny that something like this is true. Although his timeline is still too long, he writes: “there was clearly a major burst of evolutionary innovation and diversification in the first 20 million years or so of the Cambrian.”
But Miller still claims that there are Precambrian bilaterian ancestors to the Cambrian animals. He writes:
Another bilateral form that has been the subject of much recent attention is Kimberella (Figure 5). This 555 million year old fossil has been interpreted as a stem mollusk. Scratch marks found associated with Kimberella indicate that it had some form of feeding structure (though probably not a true mollusk radula) that enabled it to graze the abundant algal mats.
As we saw, Meyer mentioned the possibility that Kimberella could be a bilaterian mollusk-like organism, and indeed a member of phylum mollusca. But even this may have been too generous. According to an authoritative review by Graham Budd and Sören Jensen, Kimberella “does not possess any unequivocal derived molluscan features, and its assignment to the Mollusca or even the Bilateria must be considered to be unproven.” Despite these ambiguities, they call this organism the “the strongest case for an Ediacaran bilaterian body fossil.” Douglas Erwin notes regarding Kimberella that “this is not clearly a mollusc (Fedonkin and Waggoner’s preferred interpretation) or even a protostome.” He thinks the connection between Kimberella and bilateria is “unproven”:
Those who would assign Ediacaran fossils to protostomes and deuterostomes have generally failed to identify any diagnostic characters which unambiguously support such assignments. At present then, claims of a higher metazoan affinity must be regarded as unproven.
(Douglas Erwin, “The Origin of Bodyplans,” American Zoologist, 39: 617-629 (1999).)
More recently Michael J. Vendrasco of California State University Fullteron has also questioned Kimberella‘s connection to true mollusks known from the Cambrian explosion:
[T]he Burgess Shale, a locality with exceptional fossil preservation, reveals that at least during the middle Cambrian there also lived much larger (> 100 mm in length) soft-bodied molluscs. Strikingly well-preserved fossils of Odontogriphus showing a distinct radula, chiton-like ventral mouth, and ring of ctenidia demonstrate that this genus is a member of the total group Mollusca (Caron et al., 2006). There is at least a superficial similarity between Odontogriphus and the similar large, flat, bilaterian Kimberella from the Neoproterozoic, and Caron et al. (2006) concluded that the latter is also a stem lineage mollusc. However, aside from bilateral symmetry and oval body, there are no distinct features in common between these two fossils. The original interpretation of Kimberella (in Fendonkin and Waggoner, 1997) included a terminal mouth and proboscis, and a differentiated dorsal surface, not at all like Odontogriphus or a typical mollusc. Parkhaev (2008) seconded Fedonkin and Waggoner’s (1997) hesitation in calling Kimberella a mollusc, and Butterfield (2006) questioned the mollusc affinity of both Kimberalla and Odontogriphus. Moreover Butterfield (2008) described an apparent radula from the early Cambrian of Canada that is more similar to the radulae of modern molluscs than is the feeding apparatus of Odontogriphus. Thus, although Caron et al. (2006) inferred that the Kimberella–Odontogriphus body plan was the likely plesiomorphic (primitive) state of the Mollusca, their hypothesis lacks strong support, and this is exacerbated by the uncertain placement of Kimberella.
(Michael J. Vendrasco, “Early Evolution of Molluscs,” in Mollusks: Morphology, Behavior and Ecology, Averkii Fyodorov and Havrila Yakovlev eds., (Nova Science Publishers, 2012).)
Vendrasco goes on to classify Kimberella within “Fossil Problematica” or “forms of uncertain affinity.” If this is the “strongest case” for a Precambrian bilaterian, it’s pretty weak.
Cladistics to the Rescue
Many of Miller’s arguments, however, use cladistics to claim there are “transitional forms” showing the evolution of various Cambrian phyla. At its heart, all cladistics does is compare traits found in organisms. Those with more similar traits (called shared derived characteristics) are said to be more closely related. The method goes no deeper than that. To use it to construct phylogenetic trees (or “cladograms”), proponents of cladistics must make many assumptions.
First, cladistics assumes rather than demonstrates common ancestry; it assumes it from the outset.
Second, cladistics assumes that organisms with more similar traits are more closely related. But that assumption constantly fails when trying to construct phylogenetic trees. This is because traits often are not distributed in a tree-like manner. This forces evolutionary biologists to appeal to convergent evolution, loss of traits, and many other ad hoc explanations in order to force-fit the data into a tree. This issue is addressed in the Epilogue to Darwin’s Doubt and Chapters 4-9 of Debating Darwin’s Doubt.
Regarding the latter point, these failures become acute with regards to the origin of arthropods. Under cladistics, Miller can take any Cambrian fossil and find that it shares some (though not all) of the traits of some group and then call it “transitional.” That’s the essence of what cladistics does. But cladistics ignores unique traits that make an organism look very non-transitional (which is the case with the Cambrian oddballs like Hallucigenia or Opabinia), and the traits it doesn’t ignore often don’t fit neatly into a tree.
An ideal illustration of this problem is seen when Miller attempts to invoke lobopods and anomalocaridids as “transitional forms” to explain the evolution of arthropods. He claims that “the lobopods have become recognized as the critical link in reconstructing the assembly of the arthropod body plan,” and also cites “stem arthropods such as the anomalocarids.” In saying “stem” he means that anomalocaridids are early precursors to arthropods — “transitional forms.”
In claiming that both lobopods and anomalocaridids are closely related collateral ancestors or “transitional forms” leading to arthropods, Miller’s arguments have the same problems that Nick Matzke’s did when he argued the same thing in response to Darwin’s Doubt. (Actually, it’s more likely that in responding to Steve Meyer, Matzke directly relied upon Miller’s arguments .) As Casey Luskin explained in Debating Darwin’s Doubt, the origin of arthropods is a major problem for evolutionary biologists because they can’t construct a neat evolutionary tree that goes from lobopods and/or anomalocaridids to arthropods. Here’s what Luskin wrote in response to Matzke:
Matzke claimed that Anomalocaris and other members of its family (the anomalocaridids) are “intermediate” to arthropods. Why? Because they share some (though not all) of the derived characteristics that define arthropods. He asserts that Anomalocaris “is one of many fossils with transitional morphology between the crown-group arthropod phylum, and the next closest living crown group, Onychophora (velvet worms).” But leading authorities doubt that the characteristics supposedly linking anomalocaridids to arthropods are homologous — in other words, that they demonstrate shared ancestry.
For example, in his initial response to Meyer, Matzke posted a cladogram from David Legg et al. (2012) linking Anomalocaris to arthropods because of their similar compound eyes and cephalic limbs (limbs extending from their heads). But he failed to include the caption, which admits serious doubts about homology between these characters, noting that compound eyes “disappeared and ‘re-evolved’ a number of times,” and that “it is unclear if the arthropodized cephalic limbs of radiodontans (Anomalocaris and Hurdia) are homologous to the arthropodized trunk limbs of arthropods.” In another article from 2013, Gregory Edgecombe and Legg even suggest that anomalocaridids might be “nonarthropods” with only “convergent similarity to arthropods” since their “affinities to Arthropoda are controversial.”
Similar problems exist with regard to another supposed “transitional form” Matzke cites — the lobopods, represented today by velvet worms, of phylum Onychophora. He claims that “the arthropod and velvet-worm phyla evolved from lobopods, and lobopods contain a whole series of transitional forms showing the basics of how this happened.” Moreover, he asserts that “Anomalocaris… is one of many fossils with transitional morphology between the crown-group arthropod phylum, and the next closest living crown group, Onychophora (velvet worms).” This suggests to him that Anomalocaris was intermediate between lobopods and arthropods. Matzke further claims that “cladistic analyses reveal the order in which the characters found in living groups were acquired.” Yet lobopods and anomalocaridids are a prime example of how cladistics has failed to reveal such ordering.
If, according to Matzke, both lobopods and radiodontans (the order that includes anomalocaridids) are closely related to arthropods, which group is more closely related to arthropods? To put the question another way, what is the order in which these two groups branch off from the tree leading to arthropods? Experts disagree on this question, and their reasons for disagreeing challenge the homology of lobopod and anomalocaridid features with arthropods.
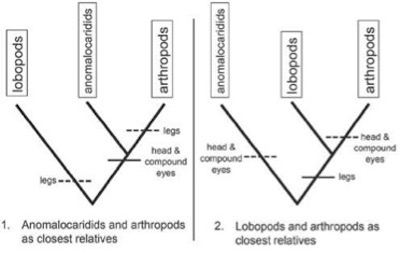
Arthropods are defined by a number of features. But understanding the origin of arthropods requires explaining how they acquired paired, jointed appendages (legs) and a complex head and compound eyes. Lobopods have legs, sometimes resembling paired and jointed appendages in arthropods, but they lack arthropod-like heads with compound eyes. Anomalocaridids lack legs entirely, but some had arthropod-like heads with compound eyes. Sharp disagreement exists over whether lobopods (with legs, not heads or compound eyes), or anomalocaridids (with heads and compound eyes, not legs), are more closely related to arthropods. Cladistic analyses have failed to yield a consensus on the branching “order” in which these crucial arthropod characters were acquired.
… Cladists must decide which is more closely related to arthropods — A or B? If you answer “A,” and the legless anomalocaridids are more closely related to arthropods, then this also means the legs of lobopods cannot be homologous to arthropod legs (and cladists have no ancestral source to explain the evolution of arthropod legs). If you answer “B,” and the compound-eyeless lobopods are closer to arthropods, then the compound eyes of anomalocaridids cannot be homologous to arthropod eyes (and cladists have no ancestral source to explain the evolution of arthropod eyes). There’s no good solution for cladists. Pick either option, and you’re faced with a situation where one of Matzke’s so-called “transitional” forms cannot be transitional, and may not even belong in the branching pattern of organisms leading to arthropods.
Figure 9-2 further illustrates this dilemma.
As seen in Figure 9-2, cladists must choose between option (1) and (2), but in either case a key arthropod trait lacks homology with another Cambrian animal group and must have evolved convergently. The solid-horizontal lines indicate an hypothesis of genuine homologies; the dotted horizontal lines would then be homoplastic (convergent) characters.
Matzke’s rebuttals to Meyer disclose none of these problems. However, a brief review of the literature shows these issues are widely recognized. A Nature article from 2011 notes that “potential stem-arthropods typically express mosaics of arthropod-like characters, which makes resolving a single, simple tree of arthropod origins problematic.” Another Nature paper frames the problem this way:
[T]he absence of sclerotized trunk limbs in dinocaridids (Anomalocaris, etc.)… remains for us one of the great puzzles of stemgroup arthropod evolution. Put simply, dinocaridids [the class which includes anomalocaridids] have a more arthropod-like head region (cephalisation, eyes, sclerotized mouthparts), whereas Diania [a lobopod] lacks such sophistication in the anterior body [head] region, but has jointed trunk appendages.
Another paper in the same exchange directly contradicts Matzke’s claim that cladistics has revealed the ordering of arthropod evolution:
The increasingly detailed fossil record of stem-group euarthropods provides our best chance of resolving this issue, but as yet has failed to do so; unequivocal evidence for any particular ordering of acquisition in these characters is not yet available.
Moreover, unique and highly derived features of anomalocaridids and lobopods make them unlikely candidates for arthropod precursors. Anomalocaris has weird flexible lobes on each side of its body, and a unique mouth with ringed teeth. As two experts explain, it “is unlike any known arthropod, particularly in the nature of the jaw apparatus and the close-spaced, strongly overlapping lateral lobes.” Lobopods have equivalent peculiarities. One paper discussing Diania, a lobopod that some have claimed is closely related to arthropods, observes that it is “a highly unusual creature. It is hard to envisage it as the progenitor of any modern arthropod group.” In constructing cladograms showing these organisms as intermediate, cladistic analysis finds it convenient simply to ignore such unique features.
Because of these problems, authorities have acknowledged that the evolutionary origin of arthropods is a real mess. Luskin further explains in Debating Darwin’s Doubt:
Multiple authorities have recognized that Matzke’s smooth evolutionary grade of intermediates leading to arthropods does not exist. Douglas Erwin and James Valentine’s 2013 tome The Cambrian Explosion, which we refer to often in this book, summarizes the debate by observing that the homology of anomalocaridid or lobopodian appendages with arthropods is one of “the problems currently facing researchers.” They write that “[t]his debate is far from settled, illustrating the complexities of understanding the evolutionary pathways among these groups.”
Likewise, Stefan Richter et al. (2013) note: “The exact composition of the stem group of arthropods depends on the position of the Onychophora and Tardigrada, which to date remains unresolved” and “there are still taxa which are the subject of debate with regard to their phylogenetic position, one being the ‘great appendage arthropods'” — e.g., the anomalocaridids.
In other words, the phylogenetic positions of both anomalocaridids and lobopods, as well as the ordering of the evolution of arthropod characters, are unestablished. Similarly, Gregory Edgecombe writes: “Arthropod phylogeny is sometimes presented as an almost hopeless puzzle wherein all possible competing hypotheses have support.” Even Matzke’s citation from Legg et al. (2012) explains: “The origin of arthropods is a contentious issue… there is little consensus regarding the details of their origins.”
One final point about cladistics, which Steve Meyer explains in the Epilogue to Darwin’s Doubt: Cladistics does not address the mechanism of evolution. It only deals with trying to show evolutionary relationships and document common ancestry, and cannot tell you anything about how traits arose. So even if all of Miller’s cladistics arguments are provisionally taken for granted, he still cannot refute Meyer’s arguments about the lack of an evolutionary mechanism to explain the origin of body plans that appear abruptly in the Cambrian. As Meyer writes in the Epilogue:
Cladistics describes patterns of relationships among organisms; it provides tools for classifying organisms. It might also suggest historical reconstructions of evolutionary history if its question-begging assumptions in that context are granted. But it cannot determine what caused the patterns of relationship depicted by cladograms or what caused the origin of the complex animal features that it analyzes. For this reason, cladistics cannot be used to rebut the central argument of Darwin’s Doubt, which addresses precisely the question of what caused the Cambrian animals to arise.
As noted, Meyer addresses cladistics extensively in the Epilogue to Darwin’s Doubt. Much of this is also addressed in the Section II of Debating Darwin’s Doubt.
Return of the Artifact Hypothesis
To explain the Cambrian explosion, Miller also invokes the artifact hypothesis: “Because of the biases of the fossil record, the most abundant and geographically widespread species of hardpart-bearing organisms would tend to be best represented.” But Meyer deals with this argument in Darwin’s Doubt, noting that the prevalence of soft-bodied fossils in the Precambrian — yet the simultaneous lack of Precambrian fossils that can serve as ancestors to Cambrian animals — shows that the artifact hypothesis does not explain the Cambrian explosion.
Thus, when Miller invokes the Precambrian Ediacaran fauna to claim, “In keeping with evolutionary expectations, fossils are now known from the late Precambrian and early Cambrian that record several dramatic transitions in the history of life,” he misstates the evidence. Meyer’s take is far more accurate. He explains that “leading paleontologists also doubt that the Cambrian animals descended from these Ediacaran forms” and concludes:
In a best case, the Ediacaran forms represent possible ancestors for, at most, four distinct Cambrian body plans, even counting those documented only by trace fossils. This leaves the vast majority of the Cambrian phyla with no apparent ancestors in the Precambrian rocks (i.e., at least nineteen of the twenty-three phyla present in the Cambrian have no representative in Precambrian strata).
Meyer further notes that authorities don’t see clear-cut Precambrian bilaterian ancestors to Cambrian animals:
As Graham Budd and Sören Jensen state, “The known [Precambrian/Cambrian] fossil record has not been misunderstood, and there are no convincing bilaterian candidates known from the fossil record until just before the beginning of the Cambrian (c. 543 Ma), even though there are plentiful sediments older than this that should reveal them.” Thus they conclude, “The expected Darwinian pattern of a deep fossil history of the bilaterians, potentially showing their gradual development, stretching hundreds of millions of years into the Precambrian, has singularly failed to materialize.”
Lastly, Miller tries to address the cause — which he calls the “trigger” — of the Cambrian explosion. He invokes a small but typical smorgasbord of familiar explanations — global Precambrian glaciation changed the temperature and chemistry of the oceans, leading to higher oxygen levels, the origin of hard parts led to an evolutionary arms race, and various feedbacks caused these factors to play off of one-another. Each of these shares the same glaring problem: None accounts for the origin of new information needed to explain the origin of the new animal body plans that appear in the Cambrian. For a discussion, see here.
In sum, Keith Miller’s white paper is a nice summary of many of the typical pop-evolutionary arguments that are invoked to explain away the Cambrian explosion. Those arguments just don’t work. He opens with bold claims of how “[i]mportant transitional steps between living phyla and their common ancestors are preserved” including “the rise of mollusks” and “the evolution of arthropods from lobopods.” But when you dig into the technical literature and read the fine print, you find that there’s a lot of disagreement about claims of Precambrian ancestral bilaterian fossils, and that many Cambrian animals don’t make very good “transitional forms” to explain the origin of the phyla.
You will also find that many mainstream authorities agree with the view, expressed by ID proponents, that there really was a unique burst of novel biodiversity in the Cambrian. It is what numerous authorities call a geologically “sudden” explosion. Miller’s “long fuse” approach, redefining the Cambrian explosion as a longer period of time, doesn’t explain that burst of novelty. Superficial explanations about “triggers” for the Cambrian explosion, or invocations of the artifact hypothesis, are woefully insufficient to explain the new biological information needed to build the animal body plans that appear abruptly in the Cambrian.
BioLogos handled the matter best when its former president Darrel Falk acknowledged a “big mystery associated with the Cambrian explosion,” namely “the rapid generation of body plans de novo.” If Keith Miller’s white paper had been so forthcoming, BioLogos readers would be better served.
Photo: Kimberella quadrata, by Aleksey Nagovitsyn (Arkhangelsk Regional Museum) [GFDL or CC BY-SA 4.0-3.0-2.5-2.0-1.0], via Wikimedia Commons.