 Intelligent Design
Intelligent Design
 Life Sciences
Life Sciences
Nanotechnology Engineers Struggle to Match Cell Performance
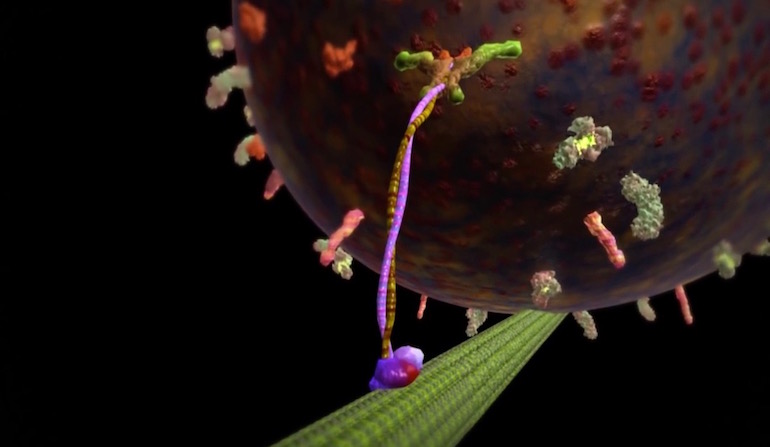
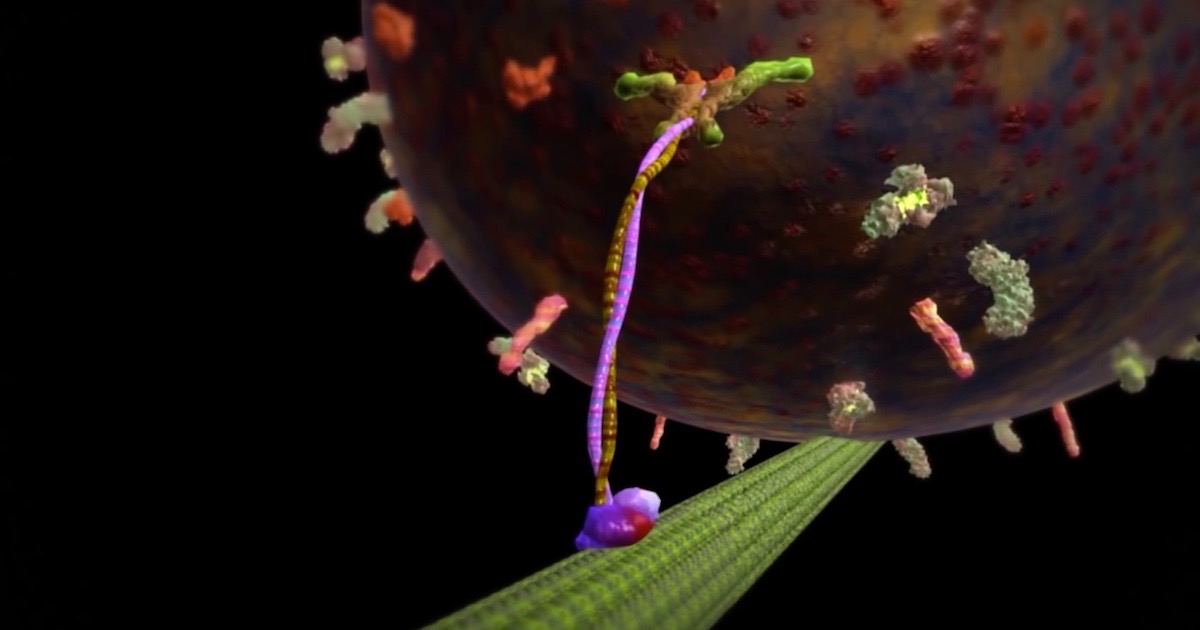
Cells make mechanical work look easy, but imitating what they do is very, very hard for intelligent designers of the human kind.
Moving Parts
Nanotechnologists are taking baby steps toward imitating what cells do all the time. In our bodies, each muscle cell uses myosin motors on actin filaments to generate tightly regulated pushes and pulls. While individually small, these physical forces added up to allow a competition weightlifter to hoist several times his body weight over his head. In other parts of our cells, kinesin motors walk along microtubules, delivering cargo where it is needed (see our animation). These moving parts are tightly regulated, directional and efficient. Now try building a molecular motor that can do these things.
In “Gearing up molecular rotary motors” in Science Magazine, Massimo Baroncini and Alberto Credi take a look at progress being made by nano-engineers trying to imitate motorized motors in cells. They have a long, long way to go.
Machines and motors based on synthetic small molecules are realized by a “bottom-up” approach to nanotechnology and could exploit molecular motion in one of two ways. The first generates macroscopic work by collecting the actions of many nanodevices organized in an array that provides spatial and temporal control of the motion activated by an energy supply. This approach mimics myosin motor proteins in skeletal muscles. The second route uses the energy-consuming directed movement of individual molecular machines to perform a task at the nanoscale, mimicking kinesin-based transport. Both cases mechanically couple an active component (the molecular machine) with nearby passive components and, ultimately, with the surrounding environment. On page 964 of this issue, Štacko et al. report the synchronous transmission of a photoactivated directional motion from a synthetic molecular motor to a coupled rotor. This demonstration takes an important step forward toward more complex mechanical functions with artificial nanoscale devices. [Emphasis added.]
Baroncini and Credi try to put a positive spin on the artificial motors, but a look at the figures and actual accomplishments in the article show the products to be pitiful contenders. One is two molecules joined by a bond that spins uncontrollably and randomly. A team made progress by adding a kind of brake to it. Another model shows a design with a molecular paddle that can be rotated in one direction with light, but what does it actually do? Very little. It was considered a major accomplishment to keep the rotating part oriented to the stator.
While we applaud every bit of progress in this very challenging arena, the real lesson is what they are learning about design requirements.
Leaving aside the stereochemical considerations required for a detailed understanding of the coupling mechanism, the key message conveyed by the study of Štacko et al. is that the transmission of motion relies on an appropriate tuning of the energy barriers associated with the different rotary motions. Another important requirement is the presence of diagnostic elements that enable the unambiguous experimental identification of the structures involved in the operation cycle. Both goals have been achieved by means of an ingenuous [sic? naïve? artless?] molecular design.
When we ask just how ingenious the design is, we finally hear an admission that cells do things far, far better than we can. The ending paragraph says:
An important feature of the present system compared with previous examples of controlled movements transferred within synthetic molecular devices is that the rotation generated by the motor is unidirectional, continuous, and autonomous (that is, it takes place under steady experimental conditions as long as light energy is available). Such extremely valuable properties are preserved upon transmission of motion. In living organisms, tasks ranging from signal transduction to motility are carried out by propagating molecular movements via mechanical connections. Although we are still far from reaching similar goals with artificial systems, the field of molecular machines is rapidly progressing, and elements now exist for taking up the challenge of making sophisticated nanoscale devices by coupling mechanical parts.
In other words, just getting a molecule to spin in one direction has been a major challenge. It’s going to take a long time before they add the signaling system and the mechanical connections to make their motors actually do some useful work.
Controlled Access
Another paper in Science explores how cells control what goes into and out of the cell membrane. Active transport is key to maintaining the cell far from equilibrium, which is part of what it means to be alive. Natural diffusion would bring everything to equilibrium, and life would stop. Nanotechnologists would like to be able to control passage of molecules, so they look to cells to learn their tricks.
What they find is an optimization problem. It’s about “Maximizing the right stuff: The trade-off between membrane permeability and selectivity,” say five researchers from Korea and America. Engineers are always looking for ways to improve filters for things like “desalination, dialysis, sterile filtration, food processing, dehydration of air and other industrial, medical, and environmental applications.” A good filter can’t be too picky or it will slow down, but it can’t be too permissive or bad things will leak through. These constraints often work against each other. For instance, how would you design a filter that could let tennis balls pass, but keep ping pong balls out? Try that for an engineering challenge! Yet some membrane channels in living cells succeed at something similar on the atomic scale:
Unlike synthetic membranes, biological membranes exhibit both high permeability and high selectivity. For example, the potassium ion channel in cell membranes is thousands of times more permeable to potassium than sodium ions, despite the smaller ionic (i.e., crystallographic) size of sodium, and exhibits permeation rates (~10^8 ions/s) approaching the diffusion limit.
One can’t just adjust the pore sizes in a filter, obviously, to achieve that kind of performance. A good filter needs other ways to discriminate between objects trying to get through. So far, though, human-designed filters that are sufficiently discriminating operate “orders of magnitude more slowly” than biological filters.
All membranes exhibit a trade-off between permeability — i.e., how fast molecules pass through a membrane material — and selectivity — i.e., to what extent the desired molecules are separated from the rest. However, biological membranes such as aquaporins and ion channels are both highly permeable and highly selective. Separation based on size difference is common, but there are other ways to either block one component or enhance transport of another through a membrane.
By imitating the selectivity filters in cells, “design features from biological membranes have been applied to break the permeability-selectivity trade-off.” Cells show that you can get your tennis balls and keep the ping pong balls out. Once again, though, humans remain far behind in this kind of engineering challenge. You almost hear a sense of awe in their jargon:
Biological membranes, such as potassium ion channels and aquaporins (Fig. 2, E and F), have extremely high selectivity-permeability combinations, which has stimulated recent efforts aimed at (i) direct incorporation of such structures into membranes, (ii) theoretical studies aimed at understanding optimal structures (Fig. 2G) that might yield high permeability and selectivity, or (iii) synthetic membrane structures that mimic or are inspired by one or more elements of biological membranes. So far, incorporation of, for example, aquaporins into membranes has been done via assimilation of aquaporins into vesicles and integration of the resulting vesicles into membranes, but there are no successful, reproducible studies demonstrating that this strategy can produce highly selective membranes. Thus, much remains uncertain about their ability to be processed into the large-scale, defect-free structures required for practical applications or whether they can maintain adequate transport and selectivity properties upon exposure to complex, real-world feed mixtures for extended periods of time.
Translation: We can’t even borrow cell technology so far, let alone imitate it. And even if we did, could it work for extended periods of time in complex, real-world situations?
The lengthy review paper describes case after case of needs banging against requirements. How can engineers use carbon nanotubes, graphene and other trendy materials to achieve cellular performance? If they could, all kinds of wonderful applications could be in our futures: better desalination plants, improved batteries, water purification, food processing, distillation, and a host of medical devices for separating blood, performing dialysis, delivering drugs and much more. Cells make it look so easy. The authors don’t have much to say about how Darwinian evolution achieved such high performance. Like, nothing.
The authors spend the last three paragraphs discussing the outlook for future progress. “Molecular-level design and insight, including advanced simulation and modeling, will be critical for breakthroughs going forward,” they say. Yet our “fundamental understanding” of filtration at the molecular level “is at an extremely rudimentary level.”
In short, the best answers will come through biomimetics: imitating how cells do it. Cells have set a very high bar. The future of science, both for theoretical understanding and application, is focused on intelligent design. Without coming out and saying the banned phrase, these papers show it. Now if they can get their molecular machines to assemble from other molecular machines following coded instructions, and to reproduce themselves, they’ll really be onto something.
Image: From “The Workhorse of the Cell: Kinesin,” via Discovery Institute.
