 Evolution
Evolution
 Intelligent Design
Intelligent Design
Protein Mutations Are Highly Coupled
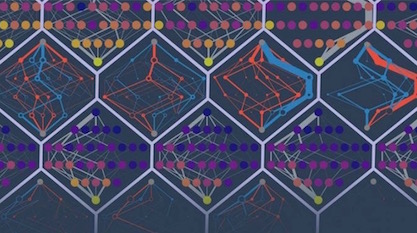
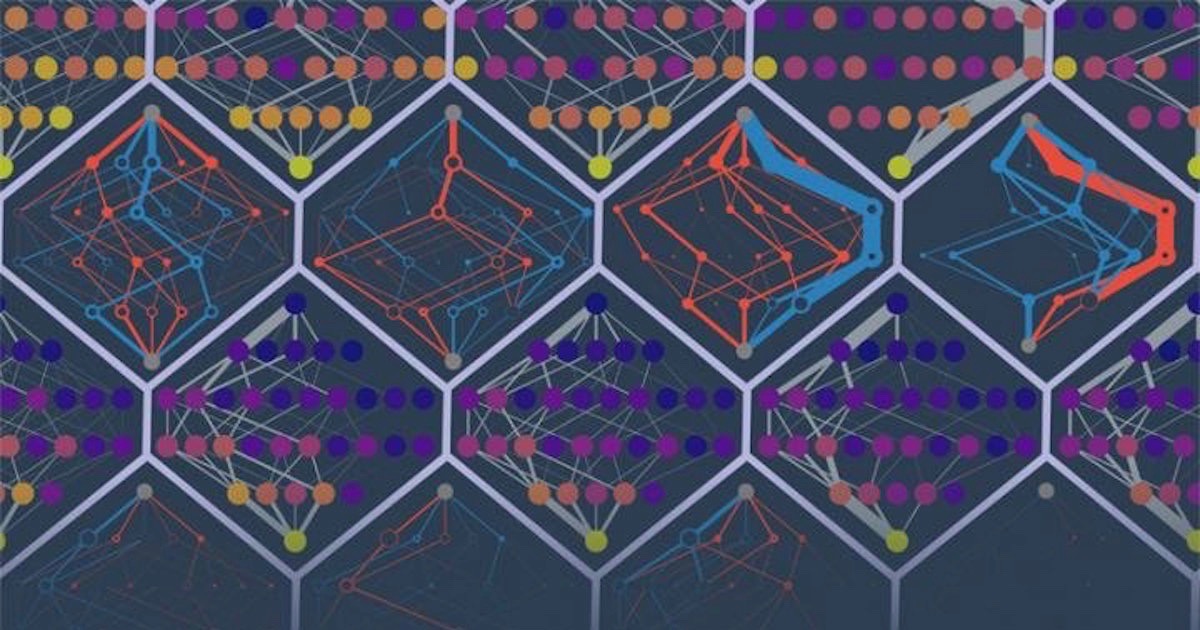
A new study from Michael Harms’s laboratory at the University of Oregon finds that potential amino acid substitutions in protein sequences are highly coupled. That is, if one residue mutates to a new amino acid, the swap impacts the other possible substitutions — they now have a different impact on the protein tertiary structure. As the paper explains:
Proteins exist as ensembles of similar conformations. The effect of a mutation depends on the relative probabilities of conformations in the ensemble, which in turn, depend on the exact amino acid sequence of the protein. Accumulating substitutions alter the relative probabilities of conformations, thereby changing the effects of future mutations. This manifests itself as subtle but pervasive high-order epistasis. Uncertainty in the effect of each mutation accumulates and undermines prediction. Because conformational ensembles are an inevitable feature of proteins, this is likely universal.
This coupling leads to a “profound unpredictability in evolution,” and the authors conclude that “detailed evolutionary predictions are not possible given the chemistry of macromolecules.”
This finding seems to confirm what many evolutionists have said for decades — that evolution is a contingent, not a law-like, process:
- “[Macro]evolutionary happenings are unique, unrepeatable, and irreversible.” — Theodosius Dobzhansky, 1957.
- “Laws and experiments are inappropriate techniques” for explaining evolutionary events and processes. – Ernst Mayr
- What science needs are “plausible scenarios for a fully material universe, even if those scenarios cannot be currently tested.” — Victor Stenger, 2004
- “Any replay of the tape would lead evolution down a pathway radically different from the road actually taken.” — Stephen Jay Gould
All of this is in direct contradiction to the science, which reveals undeniable patterns in biology that have been repeated over and over. From the pervasive instances of convergence, recurrence, and all kinds of other “ence’s”, to the non-adaptive patterns discussed by Michael Denton, the biological is anything but haphazard or random. Clearly, the same solution, for whatever reason, is used repeatedly across a wide range of species, in various patterns.
This is a clear falsification of an evolutionary expectation expressed across many years, and widely held by a consensus of experts.
But there is another problem with these protein findings. In addition to confirming the complexity and coupling of protein folding, the findings also seem to corroborate what theoretical and experimental studies have long shown, that the fitness landscape of macromolecules in general, and proteins in particular, is rugged.
The problem of evolving a protein is difficult for several reasons. First, protein function drops off rapidly with only a few mutations. Very quickly a protein loses its function as you move away from the native sequence.
Second, random or starting sequences are stuck in a flat and rugged fitness landscape. There is little sign of the kind of smooth and gradually increasing fitness landscape that would aid evolution’s enormous task of figuring out how proteins could evolve.
These problems are just getting worse, and this new finding is a good illustration of that trend.
Image credit: Michael Harms and Zach Sailer, via Science Daily/University of Oregon.
Cross-posted at Darwin’s God.
