 Evolution
Evolution
 Intelligent Design
Intelligent Design
As Research Advances, Debunking “Junk DNA” Is Almost Trendy
Jonathan Wells can smile — his 2011 book The Myth of Junk DNA remains way ahead of the curve. He writes there that “the idea that most of our DNA is junk,” even though it had been “the dominant view among biologists,” continues to show itself to be “spectacularly wrong.” Here are some recent findings that bring to light unexpected treasures in the supposed DNA junk pile.
Sex Determination Depends on Non-Coding DNA
Writing at Evolution News, David Klinghoffer noted this recent discovery from the Francis Crick Institute. The paper by Nitzan Gonen et al. in Science is here. World Magazine highlighted this story, reporting that Gonen was shocked at the impact one enhancer had. She said that some still refer to the 98 percent of our genome that doesn’t code for proteins as “junk” DNA, but her team will now be looking further for answers in those regions.
Jumping from Junk to Function
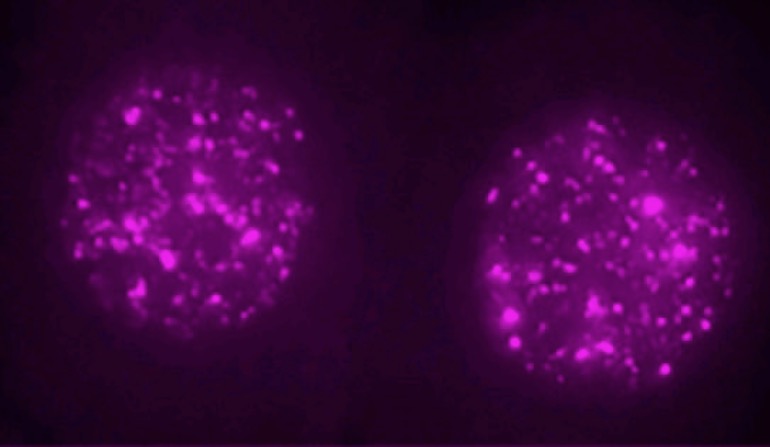
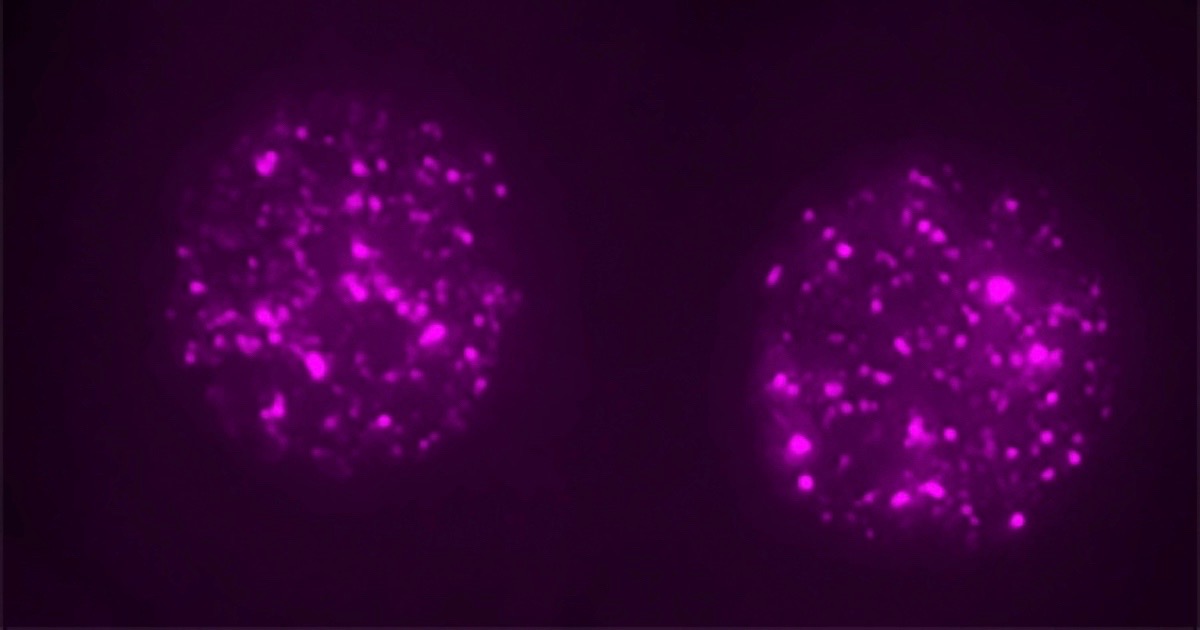
It’s almost trendy to debunk junk. From the University of California, San Francisco, comes this headline: “Not Junk: ‘Jumping Gene’ is Critical for Early Embryo.” Remember jumping genes, those alleged parasites that were taking advantage of us and evading our defenses? The subtitle of this article says, “Gene that Makes up a Quarter of the Human Genome Is Not a Parasite, but Key to the First Stages of Embryonic Development.” Nicholas Weiler explains the big turnaround in thinking, based on research with mouse embryos:
A so-called “jumping gene” that researchers long considered either genetic junk or a pernicious parasite is actually a critical regulator of the first stages of embryonic development, according to a new study in mice led by UC San Francisco scientists and published June 21, 2018 in Cell.
Only about 1 percent of the human genome encodes proteins, and researchers have long debated what the other 99 percent is good for. Many of these non–protein coding regions are known to contain important regulatory elements that orchestrate gene activity, but others are thought to be evolutionary garbage that is just too much trouble for the genome to clean up….
Now UCSF scientists have revealed that, far from being a freeloader or parasite, the most common transposon, called LINE1, which accounts for fully 24 percent of the human genome, is actually necessary for embryos to develop past the two-cell stage. [Emphasis added.]
One Controls Function, Not Junk
On the subject of jumping genes, a post by AgriLife Today talks about “Understanding How to Control ‘Jumping’ Genes.” Transposons, discovered by Barbara McClintock in the 1940s, can make up 40 percent of the human genome and 90 percent of plant genomes, according to the article. Here’s their debunk-the-junk statement:
“For years, they had been thought of as useless or ‘junk’ DNA,” Zhang said. “However, it has been recently known that transposons also play very important roles in gene regulation and evolution regardless of the potential deleterious effect.”
The partial about-face leaves some room for “potential” deleterious effects and for evolution. The actual research, though, published in Developmental Cell, shows a gene named Serrate that prevents transposon transcripts from being silenced in Arabidopsis, indicating that the transposon transcripts are vital at a certain expression level. A homolog in humans, when knocked out, is lethal to embryos. The authors perceive some elegant fine-tuning going on:
“It totally makes sense if you take a close look at the function of transposons because they are important but also harmful to the host if their expression level is too high” Ma said. “Transposon genes must be tightly controlled by balanced forces to allow low but essential expression. I’m still very much impressed by the beautiful and elegant natural design that the plant uses a single protein to fine tune the gene expression level.”
What’s a Gene, Anyway?
“Some 15 years after the human genome was sequenced, researchers still can’t agree on how many genes it contains,” says Nature, describing a new gene tally that is reigniting debate. Early estimates put the number of genes at 20,000, but that “involved tipsy geneticists, a bar in Cold Spring Harbor, New York, and pure guesswork,” Cassandra Willyard quips. A new estimate has raised the number to 21,000 — “a substantial jump from previous estimates” — including “almost 5,000 genes that haven’t previously been spotted — among them nearly 1,200 that carry instructions for making proteins.” So is that the final tally?
But many geneticists aren’t yet convinced that all the newly proposed genes will stand up to close scrutiny. Their criticisms underscore just how difficult it is to identify new genes, or even define what a gene is.
If scientists can’t define a gene, they obviously cannot define a non-gene.
Further confounding counting efforts is the imprecise and changing definition of a gene. Biologists used to see genes as sequences that code for proteins, but then it became clear that some non-coding RNA molecules have important roles in cells. Judging which are important — and should be deemed genes — is controversial, and could explain some of the discrepancies between [Steven] Salzberg’s count and others.
We’re seeing an overturn of the notion that jumping genes are “pernicious parasites” or else “evolutionary garbage that is just too much trouble for the genome to clean up.” That was an evolutionary story imposed on the data, now known to be erroneous. Design advocates would look at these unknown stretches and hypothesize, “If something exists in a working system, it must be there for a reason.” That hunch seems to be the best strategy moving forward.
Having an accurate tally of all human genes is important for efforts to uncover links between genes and disease. Uncounted genes are often ignored, even if they contain a disease-causing mutation, Salzberg says. But hastily adding genes to the master list can pose risks, too, says Frankish. A gene that turns out to be incorrect can divert geneticists’ attention away from the real problem.
Willyard raises the issue of “geneticists’ attention” and portions of the genome that “are often ignored.” Does this indicate an unconscious bias, as if their predecessors taught them to go after the shiny object (the gene), failing to see the big picture?
Function Everywhere
The concept of the gene will undoubtedly endure for some time, given its history, as long as it remains useful. But it is a human category that does not necessarily carve nature at its joints. What if the gene concept evaporates as knowledge moves forward? Why not treat the whole genome as functional? This is a radical concept, but perhaps the focus on genes (whatever they are) distorts our understanding.
We learned from ENCODE that at least 80 percent of the genome is transcribed. So what if some of it is not translated into proteins? We already know that the old “one-gene, one-enzyme” principle is wrong. Gene transcripts often undergo substantial modification by the spliceosome and by other non-coding RNA transcripts. With alternative splicing, one “gene” can generate multiple proteins. If non-coding transcripts regulate the destiny of gene transcripts, why should they get any less respect? The homeostasis of the organism is the important thing, and every worker has a role, the “grip” in the studio as well as the prima donna.
Looking back in the decline of the myth of junk DNA, we have already seen “junk” DNA playing an architectural role by providing a scaffold for chromosomes, acting like a rheostat to control transcript numbers, and working like switches to turn other genes on or off. We’ve seen “pseudogenes” (still assumed to be decaying remnants of evolutionary duplication events) acting as regulators. Even the role of “mutational sponge” can be considered a function! Where is the junk? Where are the freeloaders in this factory?
Impact on Epigenetics
A new book by Bonduriansky and Day, Extended Heredity: A New Understanding of Inheritance and Evolution (Princeton, 2018) was reviewed in Science by Kevin Laland under the title, “Beyond epigenetics.” The two book authors are “respected evolutionary biologists” who have studied nongenetic inheritance (NGI) for years. Laland says that “Weismann’s barrier” between the germ line and the somatic line has proved “remarkably porous.” The authors of this “admirable book” show, he says, that “Epigenetic inheritance is only the tip of the nongenetic iceberg.” Laland’s final paragraph sounds as if Darwinians are going to have trouble going “beyond epigenetics”:
The authors conclude that, although NGI “supplements rather than supplants genetics,” “extended heredity clearly challenges key assumptions” of neo-Darwinism and pushes us to redefine evolution as “changes in all heritable traits, whether genetic or nongenetic.” The traditionally minded may find such suggestions taxing. Convinced or not, readers will appreciate Extended Heredity as a major contribution to an exciting field.
Conclusion
In these heady days of paradigm shifts and rapid discoveries, design advocates must remain cautious about completely overhauling classical genetics to look for function everywhere, or to blend genetics with epigenetics. Harmful mutations do occur, genetic diseases are known, and even non-lethal or neutral mutations may not be optimal for the health of the organism. In addition, researchers can still make technical errors in standard procedures used to uncover epigenetic data, as a Science Daily article points out. But already, so many rehabilitations of previously considered junk have surprised biologists, we should grant the remaining suspects accused of freeloading the benefit of the doubt: “innocent until proven guilty.”
Photo credit: Ramalho-Santos lab, via University of California, San Francisco.
