 Evolution
Evolution
 Intelligent Design
Intelligent Design
Chromosomal Fusion and Correcting Mistakes: A Retrospective on an Old Debate

Over a series of articles I’ve been reviewing a 2007 lecture at the American Museum of Natural History by Eugenie Scott, titled “What Do Creationists Believe about Human Evolution?” The lecture was recently posted on the Leakey Foundation’s YouTube channel. As we’ve seen, Dr. Scott, who at the time led the National Center for Science Education (NCSE), makes the mistake of conflating ID with creationism, overstates the evidence from hominid skulls for human evolution from apelike precursors, and hinders science by prematurely (and ultimately wrongly) declaring the beta-globin pseudogene to be non-functional junk DNA.
The final argument for evolution in her lecture comes from human chromosomal fusion. As the argument goes, apes have two more chromosomes than humans (48 in apes versus 46 in humans). So if common ancestry is correct then we must account for this missing pair of homologous chromosomes in humans. Purportedly, we can do this by finding that human chromosome 2 appears to be the result of fusion between two ape chromosomes. Scott calls this “one of the very best bits of evidence for common ancestry of humans and apes.”
As in the pseudogene example, Scott is borrowing arguments from Kenneth Miller’s Kitzmiller v. Dover testimony. I responded to Miller long ago, in late 2005 or early 2006, at the IDEA Center in an article titled “And the Miller Told His Tale: Ken Miller’s Cold (Chromosomal) Fusion.” In her lecture Dr. Scott cites and critiques my article, saying it is “so full of basic misunderstandings of basic biology that it isn’t funny.”
Ignoring My Main Argument
This may seem like ancient history, but bear with me. Before we get into the specifics, it’s important to observe that Dr. Scott never even attempts a rebuttal to my main argument in the article. My main point about chromosomal fusion has never been to dispute the evidence for fusion in human chromosome 2 — in fact my article concedes for the sake of the argument that there is “good direct empirical evidence for a chromosomal fusion event which created human chromosome 2.” My main argument is also not that an individual with a fused chromosome could not spread that trait throughout a population. My purpose instead has been to rebut something like what Francis Collins wrote about human chromosomal fusion in The Language of God. As he said there, “it is very difficult to understand this [fusion] observation without postulating a common ancestor.” This is not at all the case, because humanity could have been separately designed from apes with 48 chromosomes, and then we experienced a fusion event in our history. Under this simple scenario the evidence would appear exactly as it is, as seen in the diagram below:
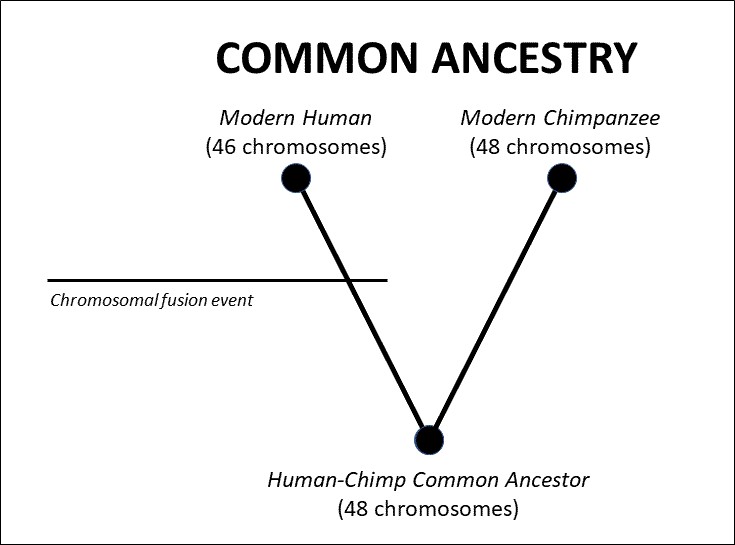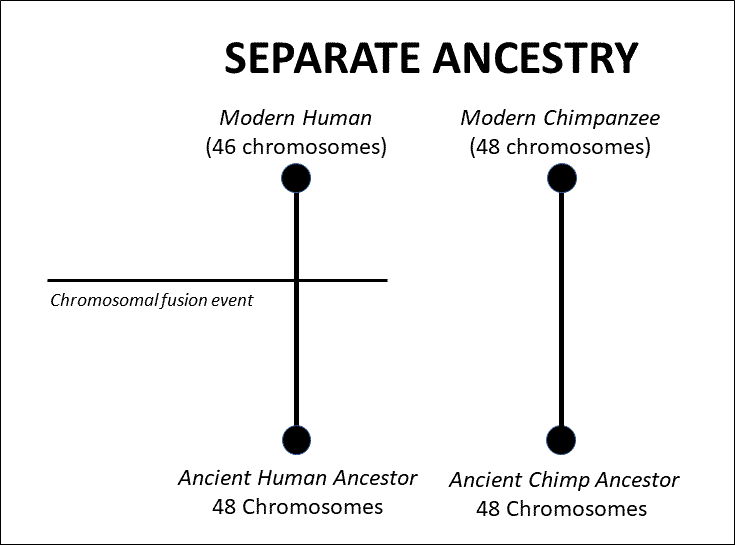
Thus, it’s very easy to account for the fusion evidence without requiring a common ancestor between humans and apes. At most what this evidence requires is that we derive from a 48-chromosome ancestor. Whether that ancestor traces back to a common ancestor we share with apes is a separate question that is not addressed by the fusion issue.
Phylogenetically Indeterminate Evidence
When considering phylogenetic relationships, it’s important to appreciate the fact that this fused chromosome is found only in humans (and not apes). In the language of cladistics, therefore, our fused chromosome is considered an apomorphy, defined by Oxford Reference as a “A novel evolutionary trait that is unique to a particular species and all its descendants.” Because this trait is found in only a single living species (humans), we would call it an autapomorphy. According to Oxford Reference, under the logic of cladistics, autapomorphies cannot provide useful information about phylogenetic relationships:
An apomorphy that is restricted to a single species is termed an autapomorphy. It alone cannot provide any information about the phylogenetic relations of that species, although it can indicate the degree of divergence of a species from its nearest relatives. An example is speech, which is found solely in humans (Homo sapiens) and not in other primates. [Emphasis added.]
The same could be said of this fusion trait: it is found only in Homo sapiens and not in other primates. Similarly, the textbook Tree Thinking: An Introduction to Phylogenetic Biology states:
These characters are uniquely derived characters (sometimes called autapomorphies). Because they can be parsimoniously explained as having evolved on a terminal branch of the tree, they do not help tell us which tips share more recent common ancestry.
p. 185, emphasis added
Likewise, the volume The Character Concept in Evolutionary Biology states:
Characters that tend to show unique states in each taxon of a clade, like any autapomorphy, will contribute little to resolving phylogenetic relationships.
p. 288, emphasis added
ID critics are quick to slam ID proponents when we make (supposed) mistakes related to cladistics or systematics. But in this situation, leading evolutionary scientists are trying to claim that an autapomorphy provides evidence of an ancestral relationship between two different types of organisms. Their own literature says this shouldn’t be done, meaning that they are trying to carve out an exception to their own rule. Scott’s lecture doesn’t touch this problem, which undercuts the import of fusion for supporting common ancestry.
I grant that there is one aspect of this evidence that provides a limited degree of evidence for common descent — namely that common descent arguably predicts something like a fused chromosome in humans given that apes have 48 chromosomes and humans have 46. In that sense this is a potential fulfilled prediction of the model,*** but it’s not special evidence for common ancestry because the same evidence is not at all hard to explain if common descent were false. Given that fact, and given that there is other evidence against common descent, I see this as a non-determinative factor in the overall debate.
(*** Note that these predictions are highly plastic. If there was no evidence for a fused chromosome in humans, evolutionists might just argue that the fusion occurred so far in the past that the evidence for it got scrambled, which is in fact exactly what they do to explain why the purported fusion site lacks much of the expected telomeric DNA. Or, they might claim that the common ancestor of humans and apes had 46 chromosomes, and that a chromosomal duplication occurred in the line [or convergently in the lines] that led to living ape species. Evolutionists tolerate huge amounts of homoplasy and other complexities in phylogenetic models, and a failure to find a fusion site would hardly trouble them about their model.)
Non-Ideological Authorities Agree
The foregoing was the main subject of the article I wrote that Scott critiques. But at the end of those 1,800 words, I added a 370-word section titled, “It only gets worse for Neo-Darwinism.” That section included a secondary argument. It was always intended to be an afterthought. But it made a point worth raising: Miller’s story implies that at some point a 48-chromosome ancestor experienced a chromosomal fusion event and then that individual interbred with others so that eventually the fused chromosome spread through the population. Yet initially, individuals with the fused chromosome would have had an abnormal number of chromosomes compared to other members of the population. And that frequently leads to problems, typically one of the following: either you aren’t viable, or you can’t produce offspring, or your offspring aren’t fertile.
Reduced to its essence, this basic point was valid, as many sources attest:
- Medlineplus.gov: “A change in the number of chromosomes can cause problems with growth, development, and function of the body’s systems.”
- Brenner’s Encyclopedia of Genetics: “Chromosome loss or gain results in massive change in DNA content that is expected to have major genetic consequences. Indeed, such mutations in the germline are often lethal. One of the most common chromosomal abnormalities in live-born children is Down syndrome, caused by trisomy (triplicate rather than the usual duplicate copies) of chromosome 21.”
- Scitable by Nature Education: “[A]neuploid cells have an abnormal number of chromosomes. Because each chromosome contains hundreds of genes, the addition or loss of even a single chromosome disrupts the existing equilibrium in cells, and in most cases, is not compatible with life. … Cells seem to be particularly sensitive to the loss of a chromosome, because the only viable human monosomy involves the X chromosome. Females with a single copy of the X chromosome have the condition known as Turner’s syndrome. Interestingly, the frequency of Turner’s syndrome is significantly lower than that of sex chromosome trisomies, suggesting that a single X chromosome is insufficient for optimum cell function. The viable Turner’s (45,X) females display a wide range of symptoms, which include infertility and impaired sexual development, and these individuals are usually mosaics.”
Textbooks and authorities on genetics thus observe that changes in the number of chromosomes frequently lead to disease, nonviability, sterility, or sterility of offspring. This suggests that left to its own course, such a fusion event might have a hard time spreading to fixation. But Eugenie Scott seems unable or unwilling to acknowledge these basic points. In fact, it was because of this very point that she said my article was “full of basic misunderstandings of basic biology” and called it “absolute nonsense.”
My Mistake
But I did make a mistake in the original article, and Scott points this out. In my original post, I said that “all of our experience with mammalian genetics tells us that such a chromosomal aberration should have resulted in a non-viable mutant, or non-viable offspring.” That was too strong a claim, because, as we’ll see, in rare situations it is possible that individuals with fused chromosomes can survive, reproduce, and in very rare cases even leave fertile offspring.
I fixed this error in my article years ago, deleting the words “all of” and “should” and changing the sentence to read:
our experience with mammalian genetics tells us that such a chromosomal aberration could have created a non-viable mutant, a normal individual who could not reproduce, or a normal individual whose offspring were infertile.
Nonetheless, both the original and fixed versions support the same conclusion: “Neo-Darwinism has a hard time explaining why such a random fusion event was somehow advantageous.”
Eugenie Scott Overstates My Overstatement
I wasn’t the only one who overstated things here. In her lecture Dr. Scott claims that I argued (her paraphrase) that “if there was a translocation [fusion] of that sort, that the animal, the organism would not be able to reproduce.” I never said anything like that. Even the original version of my article acknowledged that an individual with a fused chromosome could potentially reproduce:
In most of our experience, individuals with the randomly-fused chromosome can be normal, but it is very likely that their offspring will ultimately have a genetic disease.
I further said (in the original): “a random chromosomal fusion event … in our experience ultimately results in offspring with genetic diseases.” Now that language does wrongly suggest that chromosomal fusion must “ultimately” result in a genetic disease (I’ve also fixed this language). But it does not deny that an individual with a fused chromosome can reproduce.
I never denied that an individual with fused chromosomes can survive and even leave offspring. But its offspring may have severe disabilities, as is the case with Translocation Down’s Syndrome (where a parent has a fused chromosome) and other genetic abnormalities.
Fusion in Horses
In her lecture, the main evidence that Dr. Scott cites to argue that chromosomal fusions aren’t necessarily deleterious comes not from humans but from horses. A rare and endangered breed from Mongolia called Przewalski’s horse (Equus przewalskii) has 66 chromosomes — two more than the common domestic horse (Equus ferus caballus), which has 64 chromosomes. It has been proposed that the domestic horse arose from a 66-chromosome ancestor like Przewalski’s horse that underwent a fusion of two chromosomes during meiosis. There’s good evidence for this hypothesis: these two breeds of horse can still mate and produce fertile offspring.
Scott’s claim about horses traces back to a 1974 article by Short et al. in the journal Cytogenetics and Cell Genetics, “Meiosis in interspecific equine hybrids.” They proposed a potential genetic mechanism by which a chromosome could have been fused in Przewalski’s horse, which then could have spread through a subpopulation via genetic drift. The key is that all of the original genetic material is there — it’s just been moved around (translocated) as two chromosomes became fused. Whether or not something like this happened in humans, it’s not impossible for a fusion of this type to become spread in a population.
But as to my updated argument, this paper supports my point. There are at least seven different living species of equines, and they all have different numbers of chromosomes. The paper has a very useful table showing what happens when those different species of horses attempt to interbreed:

Of all known cases of attempted cross-breeding between these seven living species, in only one example can the mating lead to fertile offspring. This is the one that Eugenie Scott cites — a fertile hybrid between Przewalski’s horse and the domestic horse. In all the other cases you either get no offspring (8 cases, it would seem) or sterile offspring (12 cases). That’s pretty interesting. In the best case that Eugenie Scott raises — horses — only 1 in 21 types of potential hybrids between parents of different chromosome numbers is known to produce fertile offspring. This shows it’s not impossible for a fusion event to be spread throughout a population, but it also supports my general point that changes in or differences to chromosome numbers are frequently barriers to evolutionary success.
Missing Obscure Literature
Should I have known about this obscure paper from 1974? Ideally, sure. But there’s a lot of literature out there. And I’m not the only one here who has missed literature.
The hypothesis that the beta-globin pseudogene (HBBP1) is functional, which I discussed in my previous response, also existed long before Scott and Miller declared it was a “non-functional” broken gene to courts, the media, and the public. The 2013 paper which first identified empirical evidence supporting functionality for this pseudogene makes a relevant passing comment:
Several decades ago, a hypothesis was formulated holding an important regulatory role of HBD and HBBP1 in the Hb fetal-to-adult switch that matches quite well the assumption of strong negative selective forces acting on these sequences (Ottolenghi et al. 1979; Bank et al. 1980; Chang and Slightom 1984; Goodman et al. 1984).
So, papers from the 1980s had already proposed that the beta-globin pseudogene is functional and has a regulatory role. I dug up those citations and found that one of them from 1984 clearly stated a hypothesis that the beta-globin pseudogene (often called Ψ-globin) appears to have a conserved sequence, has a function to regulate other protein-coding beta-globin genes, and is involved in the switch from fetal to adult globin expression. Here’s exactly what was proposed by Chang and Slightom (1984) in the Journal of Molecular Biology:
Our analysis of the ß-globin gene family of gorilla and chimpanzee, by lambda cloning followed by restriction mapping, blot hybridizations, and DNA sequencing clearly shows that these primate species have the same organization of ß-globin genes, including the pseudogene, as that found for humans. The Ψß-genes of the three hominoids contain the same transcriptional and translational defects, they were clearly pseudogenes before the three species separated. The accompanying papers by Harris et al. (1984) and Goodman et al. (1984) confirm that this gene locus has a very long evolutionary history.
Reasons why this locus has been conserved are not yet apparent. Possibly after its inactivation its role may have changed from that of a polypeptide-encoding gene to that of regulation of other genes in the ß-globin gene family. Fritsch et al. (1979), Bernards & Flavell (1980) and Collins & Weissman (1984) have suggested that DNA sequences located between the y- and b-genes may be important in maintaining a cis-acting regulatory signal necessary for the switch from fetal to adult globin gene expression, which occurs shortly before birth. Such a cis-acting regulator may not directly involve specific sequences in the Ψß-gene locus, but the presence of this locus may be necessary for the proper phasing of the regulatory mechanism. In this connection, our major finding is that the stretch of DNA which constitutes the Ψß-globin locus is evolving at a rate much slower than that expected for non-functional DNA. Indeed, these hominoid Ψß-gene sequences are evolving even slower than the IVS 2 sequences for the human and gorilla fetal globin genes. With our evidence showing that the nucleotide sequences in and around the Ψß-globin locus are conserved in hominoids, the suggestion that this locus is involved in the regulation of other ß-globin genes may have merit.
This 1984 paper was prescient and turned out to be basically correct. The hypothesis, albeit stated in a relatively obscure paper, was out there in 2005 when Ken Miller testified at Dover, and in 2007 when Eugenie Scott gave her lecture claiming the Ψ-globin was “non-functional” and didn’t do “diddly.”
Reflections on the Past and Present
Mistakes happen. That’s OK. What’s important is what you do about that. I’ve corrected my error. I hope Dr. Scott does the same with hers.
In her lecture, though, she goes much further than merely critiquing a mistake. She uses it as an occasion to smear an entire class of people, stating:
Creationists excel at misunderstanding science, misunderstanding its conclusions, and getting things wrong.
The Darwin lobby has a long history of such broad-brushing and stereotyping, and they’ve never really reformed their ways. Back in 2009 I shared the stage at a symposium with an NCSE-affiliated theologian, Peter Hess, who said, “A third problem with intelligent design is that its practitioners are either ignorant of science or seriously deluded or fundamentally dishonest.” Scott herself is famous for having stated, “There are no weaknesses in the theory of evolution”—a blatant overstatement and falsehood. Statements like this are still made today. This is not ancient history at all.
Yet should we say that Scott and Miller have “basic misunderstandings of basic biology” simply because they weren’t aware of an obscure paper from the 1980s? No, that would be unfair. Should we indict them and say they “excel at misunderstanding science, misunderstanding its conclusions, and getting things wrong” simply because they made an inaccurate evolutionary prediction about a pseudogene being “non-functional”? Nope. That would be uncharitable and wrong.
Speaking about evolution, Eugenie Scott closes her talk by saying, “How sad that students are not being able to learn this wonderful, fascinating science.” She implies that we are among those who “don’t want evolution to be taught.” Then, as now, her statement is false.
Students are learning about evolution, and Discovery Institute and proponents of intelligent design do want evolution to be taught. What we don’t want are dumbed-down versions of evolution that pretend there are no “weaknesses” in the theory or use oversimplified proofs of evolutionary theory that don’t hold up to scrutiny. To my knowledge, Scott has never backed down from her “no weaknesses” claim. That, like her uncorrected smears from 2007, is a legacy that hurts this debate. But it doesn’t have to be that way. As I hope this episode has shown, it’s not too late to correct past mistakes.
