Intelligent Design
Intelligent Design
Galloping Flagella and Cilia Railroads — Getting Ready to Celebrate Twenty Years of Darwin’s Black Box
We’re approaching the twenty-year anniversary of the publication of Darwin’s Black Box (it appeared on August 2, 1996, to be precise). Michael Behe’s book inspired a generation of Darwin discontents and showed that intelligent design was (and still is) firmly based on cutting-edge science. Among other highlights, he showcased two cellular organelles to illustrate his concept of irreducible complexity: the cilium and the flagellum.
These organelles, protruding from the cell into the environment, could not be built by a Darwinian mechanism, he argued, because they are composed of multiple, independent parts required for function. Like a mousetrap, they could not work unless all the parts were present together at the same time.
All true, but in the intervening years molecular biologists have learned a great deal more about cilia and flagella. The revelations keep coming, like down to the day before yesterday.
Galloping Flagella
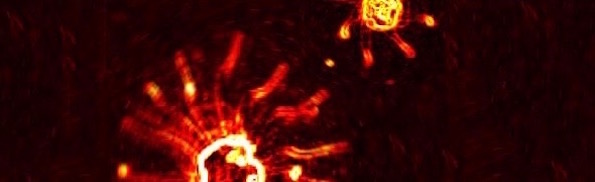
In fascinating news from the University of Cambridge, we learn that “Algae use their ‘tails’ to gallop and trot like quadrupeds.” (Those tails are Behe’s famous flagella, though differing in structure from the bacterial flagella.) You can see for yourself in the embedded videos that single-celled algae named Chlamydomonas use their flagella in coordinated ways “to achieve a remarkable diversity of swimming gaits.”
When it comes to four-legged animals such as cats, horses and deer, or even humans, the concept of a gait is familiar, but what about unicellular green algae with multiple limb-like flagella? The latest discovery, published in the journal Proceedings of the National Academy of Sciences, shows that despite their simplicity, microalgae can coordinate their flagella into leaping, trotting or galloping gaits just as well. [Emphasis added.]
They trot. They gallop. They leap in a manner similar to the “pronk” of a springbok, where the animal arches its back and springs all four feet off the ground together. The resemblance is uncanny, despite a difference in size of many orders of magnitude. The cells even know two different galloping gaits: a rotary gait and a transverse gait. How is this possible for single-celled organisms lacking neurons?
In vertebrates, gaits are controlled by central pattern generators, which can be thought of as networks of neural oscillators that coordinate output. Depending on the interaction between these oscillators, specific rhythms are produced, which, mathematically speaking, exhibit certain spatiotemporal symmetries. In other words, the gait doesn’t change when one leg is swapped with another — perhaps at a different point in time, say a quarter-cycle or half-cycle later.
It turns out the same symmetries also characterise the swimming gaits of microalgae, which are far too simple to have neurons. For instance, microalgae with four flagella in various possible configurations can trot, pronk or gallop, depending on the species.
Dr. Kirsty Wan was amazed when she first observed this, saying, “I realised immediately that this behaviour could only be due to something inside the cell rather than passive hydrodynamics.” Think of how this expands the argument for irreducible complexity:
The researchers determined that it is in fact the networks of elastic fibres which connect the flagella deep within the cell that coordinate these diverse gaits. In the simplest case of Chlamydomonas, which swims a breaststroke with two flagella, absence of a particular fibre between the flagella leads to uncoordinated beating. Furthermore, deliberately preventing the beating of one flagellum in an alga with four flagella has zero effect on the sequence of beating in the remainder.
Dr. Behe will be pleased to see this additional level of design. “The findings also raise intriguing questions about the evolution of the control of peripheral appendages, which must have arisen in the first instance in these primitive microorganisms,” one of the paper’s authors comments. Surely the first question should be, “Did it evolve at all?”
Two-Lane Highways in the Cilium
Any freshman confronted with an electron micrograph of a cilium must be impressed by the beautiful symmetry of its 9×2 structure (see the figure in Behe, p. 60). The elements of the structure are microtubules: long filaments that look like white dots in cross section. They are arranged in pairs around the periphery. The question is, “Why”? Until now, scientists were not sure. Science Magazine has just now come out with the answer: “Microtubule doublets are double-track railways for intraflagellar transport trains.” One microtubule is for going up; the other is for coming down!
Cilia contain a well-ordered array of microtubule doublets along their length. A longstanding question in cilium structure and function is why the microtubule arrangement in cilia is so complex. Stepanek and Pigino developed a time-resolved correlative fluorescence and three-dimensional electron microscopy method to show that the doublets provide directionality to intraflagellar transport. One microtubule in the pair moves cargoes up to the ciliary tip. Meanwhile, the other microtubule moves cargoes back to the cell body. These results explain why the axoneme is built out of microtubule doublets and suggest a mechanistic picture of how the logistics of bidirectional intraflagellar transport are regulated.
What’s really interesting about the paper is the constant use of railroad terminology. There are “cargo trains” with “motors” that ride on microtubule “tracks” for “transportation.” They function as “railways for intraflagellar transport” (IFT). Nowhere is a discussion of evolution to be found. Maybe that’s because everyone knows train transportation systems are intelligently designed.
Ludek Stepanek and Gaia Pigino from the Max Planck Institute observed the trains going up and down, but never colliding. They wondered how the trains avoid collisions; do they swing around each other, like using a passing lane? No; it turns out that they each have dedicated lanes.
Moreover, the upward-moving (anterograde) cargo trains use kinesin-II motors, the downward-moving (retrograde) cargo trains use dynein motors. How does each one know the lane to use, since the microtubules are made of the same track material? It appears there may be post-translational modifications of the track proteins (tubulins) that allow the engines to recognize the right lane to use. The situation gets even more complex from there:
This might contribute to the efficiency of IFT by providing optimized tracks for kinesin and dynein motors in addition to the IFT train segregation. Nevertheless, the effect of such modifications on the control of IFT in vivo remains unclear. Alternatively, a specific molecular machinery to direct trains to the correct microtubule could be present at the base and tip of the axoneme. More complex motor regulation could be necessary in the sensory cilia of Caenorhabditis elegans, where the B-microtubules do not extend beyond the middle segment of the flagellum. There, the kinesin-II moves only in the middle segment, and an additional motor, OSM-3, is required to bring cargo to the tip along the A-microtubules. Regardless of the mechanisms involved in the recognition of the microtubules by the motors, our work highlights the critical role played by microtubule doublets in the assembly of cilia.
Cilia are present on most eukaryotic cells and are involved in many essential functions, such as clearing the throat of debris (in which they wave in coordinated sweeping motions), and sensing the environment like antennae. Defects in cilium construction cause debilitating diseases or death, underscoring Behe’s concept of irreducible complexity.
It’s satisfying to report these new discoveries about cilia and flagella, confirming, as they do, Behe’s original insights. The case for intelligent design gets stronger all the time.
Image credit: Kirsty Wan and Raymond Goldstein via University of Cambridge.