 Intelligent Design
Intelligent Design
 Life Sciences
Life Sciences
An Icon Revisited: Flagellar Hook Shows Further Aspects of Design
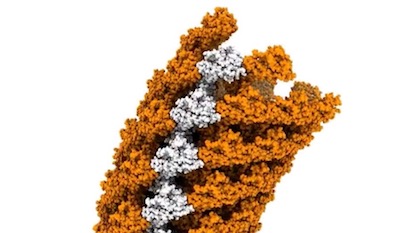
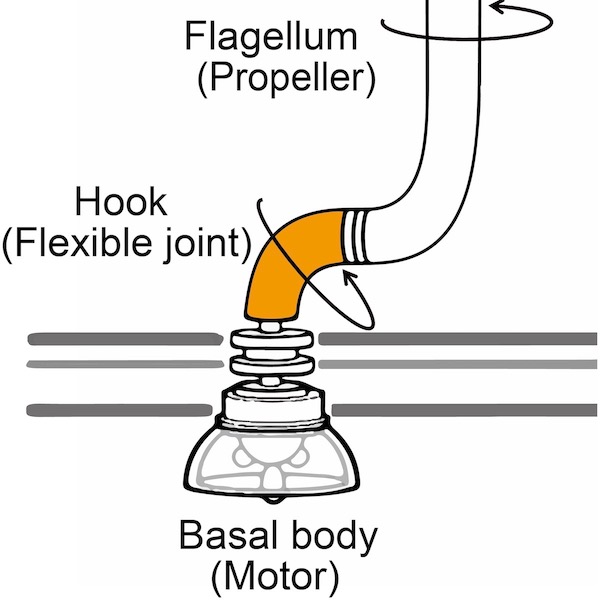
Our good old favorite ID icon, the bacterial flagellum, has not exhausted its design tricks. Closer views of the hook region show a remarkable network of linked protein components that is flexible, thereby transferring torque efficiently from the motor to the filament.
Here is an object where a picture is worth a thousand words. In an animation from a paper in Nature Structural & Molecular Biology, reproduced here by the Okinawa Institute of Science and Technology (OIST), one can see at a glance a wonderful apparatus in motion. If you tried to build a pipe that had to rotate on its long axis while maintaining its angular shape, could you do better than this?
This is the hook region of the famous bacterial flagellum that Michael Behe showcased to the world twenty years ago in Darwin’s Black Box. New methods of imaging like cryo-electron microscopy have allowed molecular biologists to see structures at near atomic resolution. Zooming in on the flagellum at such a scale, this view of the hook surpasses expectations of even ID advocates.
An Icon Revisited
In “Torque transmission mechanism of the curved bacterial flagellar hook revealed by cryo-EM,” four scientists describe what they found:
Bacterial locomotion by rotating flagella is achieved through the hook, which transmits torque from the motor to the filament. The hook is a tubular structure composed of a single type of protein, yet it adopts a curved shape. To perform its function, it must be simultaneously flexible and torsionally rigid. The molecular mechanism by which chemically identical subunits form such a dynamic structure is unknown. Here, we show the complete structure of the hook from Salmonella enterica in its supercoiled ‘curved’ state, at 2.9 Å resolution. Subunits in the curved hook are grouped into 11 distinctive conformations, each shared along 11 protofilaments. The domains of the elongated hook subunit behave as rigid bodies connected by two hinge regions. The reconstituted model demonstrates how identical subunits can dynamically change conformation by physical interactions while bending. [Emphasis added.]
The entire hook is composed of just one protein type, called FliC. The arrangement of the proteins, though, is highly sophisticated, involving precise spacing of the proteins and hinge domains between them that allow them to flex. If you remember those old Chinese finger traps childhood friends would tease us with, they’re a bit like that: evenly spaced around a hollow core, but flexible. Imagine a Chinese finger trap made of beads with hinges holding them together. It would allow you to bend the tube at an angle and roll it with the fingers, keeping the angle intact. Because of the hinges, beads approaching the outer side of the angle would spread apart as they make the turn, then compress as they approach the inner side of the angle.
Simultaneously Rigid and Flexible
The new model derived from the observations shows how the hook can be both rigid and flexible at the same time. Old “two-state” models could not account for this. The team found no fewer than 11 conformations as the proteins rotate around the helical tube.
The bacterial flagellar hook acts as a universal joint to transmit torque to filaments, which work together in a bundle pointed in a certain direction. Both the filament and the hook are helical tubes…. Based on our structure of the curved hook, it is clear that the two-state model is not applicable to the hook, because we observed at least 11 distinct subunit conformations, suggesting that there is no predetermined conformation. Instead, each subunit undergoes continuous transitions between several conformations by coordinated domain motions in each subunit.
Coordinated… work together… Those words convey irreducible complexity (IC) in a structure made of one building block. IC does not require that each element be different, as in a mousetrap. A stone arch is one example. An organized row of dominoes is another. Since the role of the hook is to transmit torque to the filament in a certain direction, is there an advantage to this hinged arrangement?
These findings demonstrate how chemically identical subunits can dynamically change conformations through physical interactions while bending, and how they transmit torque through a curved tubular structure. Our data explain a molecular mechanism for supercoiling of tubular structures and provide a rational basis for understanding allosteric transitions between polymorphic states of superhelical assemblies.
The hook needs to be hollow, because proteins for the filament are transported through the interior all the way to the tip of the filament. Although the team from OIST didn’t measure the efficiency of the transmission, they believe that “These interactions between the tightly packed spoke-like subunits convey torsional rigidity and must be responsible for transmitting most of the torque generated by the bacterial motor.” They also believe that “This geometry, together with the cooperative synchronization of the two hinge regions on each subunit via inter-protofilament links by the D2 domains, can achieve the rotational mode required for efficient torque transmission as a flexible joint.”
Flagellum Evolution
Warning: do not drink hot coffee when reading what they say about this “amazing nanomachine,” as another paper calls it. Shoving evidence and logic aside, they credit the “example of natural design ingenuity” to chance: “This intricate system is a testament to evolution’s ability to optimize over millions of years,” according to Professor Matthias Wolf.
That other paper is a preprint in bioRxiv titled, “Bacterial flagellar motor PL-ring disassembly Sub-complexes are widespread and ancient.” In this paper, Kaplan et al. find convergent evolution in a trivial fact that the P and L rings of flagella remain in the membrane when the flagellum disassembles. Is that such a big deal? “The bacterial flagellar motor is one of the most famous macromolecular machines, made up of thousands of protein subunits that self-assemble in a highly-synchronized manner into a motor, a flexible hook, and a long extracellular filament that rotates in a propeller-like fashion to move the cell,” they say. That sounds like something Michael Behe would affirm. What’s evolution got to do with it? Determined to rescue Darwin, they make up an evolutionary story to account for this “amazing nanomachine.”
An important step in reconstructing the evolutionary history of biomolecular complexes is to know when certain features and functions originated. Recent studies indicate that the bacterial flagellum is an ancient machine that originated from a single or few proteins through multiple gene duplication and diversification events that proceeds the common ancestor of bacteria. Some parts of the flagellar motor are homologous to other sub-complexes present in other machines. The stator proteins MotA/B are homologous to proteins in the Tol-pal and TonB systems while the motor’s ATPase is homologous to the beta subunit of the ATP synthase. This suggests that other, even older machines donated features and functions to the first motor. Moreover, the Type III secretion system (T3SS), also known as the injectisome, is homologous to the bacterial flagellar motor (though the P- and L-rings of the motor are not homologous to the secretin part of the injectisome). Because motility proceeded [sic] the evolution of eukaryotic cells, the targets of T3SS, and the T3SS is restricted mainly to proteobacteria, the T3SS likely derived from the flagellum.
At least they got the fact right that the T3SS is not a transitional form to the flagellum. To believe this story, one has to envision dumb molecules acting as intelligent agents, “donating” functions to one another. One has to accept that gross similarities, like rotation, link very different motors by common ancestry. Worse, one has to envision the most complex thing arising first! What is it that makes some scientists obsessed with linking disparate biological entities with ancestry? Are bicycles and motorcycles products of convergent evolution because they have two wheels? Are toy wagons and sedans related by common ancestry? Without using intelligence as a cause, such speculations make no sense.
Image credit: Okinawa Institute of Science and Technology, via EurekAlert!
