 Evolution
Evolution
 Intelligent Design
Intelligent Design
 Life Sciences
Life Sciences
PNAS Authors Resort to Teleological Language in Failed Attempt to Explain Evolution of Irreducible Complexity
Summary: A recent article in Proceedings of the National Academy of Sciences (PNAS) purports to explain the evolution of a relatively small molecular machine in the mitochondria that transports proteins across a membrane, thereby allegedly refuting irreducible complexity. Phrases and assertions like “‘pre-adaptation’ to bacteria ahead of a need for protein import,” “parts accumulate until they’re ready to snap together,” “machineries emerge before there’s a need for them,” or intelligently “engineered” macromutations are part and parcel of this latest failed attempt by critics of intelligent design (ID) to answer Michael Behe’s argument of irreducible complexity. As would be expected, when evolutionists are forced to resort to such goal-directed and teleological language and mechanisms, this shows that inherently, blind and unguided materialist explanations are not sufficient to produce irreducible complex systems. As discussed in more detail below, this latest attempt to answer irreducible complexity unwittingly shows the need for intelligent design. A summary of the problems includes:
- No ID proponent ever claimed this particular machine is irreducibly complex, so theirs is something of a straw man rebuttal.
- Most of the argument is based upon mere evidence of sequence homology with no detailed discussion of how to actually evolve bacterial proteins into the eukaryotic mitochondrial transport machine’s proteins, or solve other problems necessary to evolve this biomolecular machine.
- Most questions about the evolutionary pathway were solved by appealing to teleological-sounding explanations that the proteins were magically “preadapted” for use in the machine or that “machineries emerge before there’s a need for them.”
- In their one attempt to use a protein homologue from bacteria in the eukaryotic system to test for compatibility, the investigators could only make the protein function after they intelligently “engineered” (their word) macromutations in the protein by adding extensive key signaling sequences as well as other apparently necessary changes.
- There was no evidence found of a reduced version of this transport machine (“TIM”), so they had to try to find a reduced version of a different transport machine, “TOM” to provide “proof of principle’,” but no prokaryotic homologues for TOM were reported.
- The allegedly simpler version of the TOM transport machine still requires multiple proteins to function, allowing it to still perform all essential functions, and the system has at least two TOM proteins–not just one as the PNAS paper implies.
- The allegedly simpler TOM system was claimed to be reduced only after a genomic study, which the investigators admit would miss proteins that were performing necessary functions but were too different from previously known TOM proteins to be detected.
- According to the standard phylogeny, the allegedly simpler precursor could not even represent an ancestral form because it is descended from an ancestor that had the typical mitochondrial transport mechanisms.
Introduction
Articles on SoftPedia and Wired are touting a recent paper in PNAS titled “The reducible complexity of a mitochondrial molecular machine,” claiming it explains the evolution of irreducible complexity. The PNAS article’s opening paragraph cites to Michael Behe, stating that “Proponents of Intelligent Design have argued that these sophisticated machines are ‘irreducibly complex,’ with this standing as the proof that, at the molecular level, Darwin’s principles of evolution cannot explain the complexity of living systems.” Of course, ID critics constantly tell us there’s not supposed to be any scientific debate on the question of irreducible complexity, but never mind that.
Let’s start with the Softpedia’s article’s claims about the flagellum that the “basic building blocks of this ‘instrument’ can be found elsewhere in the cell as well, but performing different tasks.” For one, all flagellar parts do not reside “elsewhere in the cell,” as a number of necessary structures in flagella lack homologues outside of the flagellum. And even if all the parts did reside somewhere in the cell, just waiting to be co-opted for use in a functional flagellum, this would solve very little for the evolutionist. Angus Menuge explains why these co-option (also called “exaptation”) stories are weak:
For a working flagellum to be built by exaptation, the five following conditions would all have to be met:
C1: Availability. Among the parts available for recruitment to form the flagellum, there would need to be ones capable of performing the highly specialized tasks of paddle, rotor, and motor, even though all of these items serve some other function or no function.
C2: Synchronization. The availability of these parts would have to be synchronized so that at some point, either individually or in combination, they are all available at the same time.
C3: Localization. The selected parts must all be made available at the same ‘construction site,’ perhaps not simultaneously but certainly at the time they are needed.
C4: Coordination. The parts must be coordinated in just the right way: even if all of the parts of a flagellum are available at the right time, it is clear that the majority of ways of assembling them will be non-functional or irrelevant.
C5: Interface compatibility. The parts must be mutually compatible, that is, ‘well-matched’ and capable of properly ‘interacting’: even if a paddle, rotor, and motor are put together in the right order, they also need to interface correctly.
(Angus Menuge, Agents Under Fire: Materialism and the Rationality of Science, pgs. 104-105 (Rowman & Littlefield, 2004).)
Those who purport to explain flagellar evolution almost always only address C1 and ignore C2-C5. As will be seen below, this same critique applies with equal force to present arguments from this PNAS paper discussing the origin of mechanisms that transport proteins across the mitochondrial membrane.
The Refutation of a Straw Man
Mitochondria are a type of organelle in eukaryotic cells that produce ATP, a molecule that effectively carries usable energy for cellular reactions. Mitochondria are thus often termed the “batteries” or “powerhouses” of the cell. They are bounded by both an outer and inner membrane. To get a protein from outside the mitochondria to the inside of the mitochondria, you must pass the protein through both membranes through various types of transport mechanisms on each membrane. The function of these transport mechanisms is thus to grab a protein and pass it through a membrane. It’s a modest function, but an important one.
Basically, the authors of the PNAS paper tried to explain how a molecular machine that transports proteins across the inner membrane of mitochondria, called TIM23, evolved. No ID proponent has ever claimed that this particular system is irreducibly complex, making this a straw man attack. But never mind that, right?
Does “Preadaptation” and Intelligently “Engineered” Macromutations Solve C2-C5?
According to the authors of the PNAS paper, the primary evidence for the evolution of TIM23 is mere similarity between its components and other proteins that are found in bacteria. In a key summary of their argument which only solves C1, the authors state:
We proposed that simple ”core” machines were established in the first eukaryotes by drawing on preexisting bacterial proteins that had previously provided distinct functions. Subsequently, and in a step-wise process in keeping with Darwinian evolution, additional modules would have been added to the core machines to enhance their function. This proposition is supported by 3 findings: (i) that protein components found in bacteria are related in sequence to the components of mitochondrial protein transport machines, but (ii) that these bacterial proteins are not found as part of protein transport machines and (iii) that some apparently ”primitive” organisms found today have protein transport machines that function with only one or few component parts.
As can be seen, their argument primarily relies upon finding homologues of mitochondrial transport proteins elsewhere in biology. So have they explained how mitochondrial transport mechanisms evolved? Hardly. This only accounts for C1. But to truly offer “a step-wise process in keeping with Darwinian evolution,” they must account for C2-C5 and explain how a functional advantage was gained along each small step of that evolution.
Regarding these homologues, the PNAS study found that 2 proteins in TIM23 — Tim44 and Tim14 — are similar to proteins in bacteria, TimA and TimB. TimA and TimB have a different function in bacteria (bacteria don’t have mitochondria), but they do reside in the bacterial cell membrane, something like how Tim44 and Tim14 reside in the mitochondrial inner membrane. It’s suggested that if TimA and TimB spontaneously combined with a LivH amino acid transporter machine, one could evolve a TIM23 transporter complex. Here’s a summary of the steps they propose:
- Evolve a binding interaction between TimA and LivH to allow a docking point for bacterial protein Hsp70, a protein transport motor.
- Then evolve interactions between TimB and LivH.
- Then, badabing: “With these 3 bacterial proteins [TimA, TimB, and LivH] cooperating as subunits of a primitive transport machine, a step-wise evolution of the more sophisticated mitochondrial TIM complex would be enabled.”
The only real aspect of Menuge’s required explanatory components offered for all three proteins is C1. They claim that if 3 parts are lying around (step C1 from Menuge), then they’ve effectively solved the evolutionary problem. Not so. What are the odds of the parts suddenly coming together to form a functional mitochondrial transport mechanism? What are the odds of suddenly producing functional binding sites between all of these proteins? These are major problems, but they barely tried to address these questions. For the most part, they just address C1 and claim the debate is over. Their argument is like saying that if you put all of (or in this case, some of) the parts for a computer in a box, and even shake it up a little, you get a computer. But real machines require a lot more than having their parts lying around in a box.
There is a way to determine if C2-C5 pose a problem for the evolution of these proteins. They could replace the DNA encoding Tim44 and Tim14 in a eukaryotic cell’s DNA with the genes encoding TimA and TimB, respectively, and see what happens.
The authors didn’t do this for TimA (they just made sequence and structural comparisons) and thus really gave no detailed analysis of how TimA might evolve into Tim44. Nonetheless, they assert that “Point mutations in a short segment required for interaction of the TimA protein with LivH would provide a docking point for the bacterial Hsp70, which is the direct homologue of the protein transport motor,” even though they don’t identify what those point mutations would be, or how many there would need to be, or what other mutations (such as insertion of the proper signaling sequences) would be necessary to evolve a TimA-like protein to function as Tim44.
For TimB evolving into Tim14, their work is far more amusing. The authors “engineered” (their word) a modified version of TimB, by inducing mutations, not the least of which was the fact that TimB was “engineered for expression in yeast by adding an N-terminal mitochondrial targeting sequence and transmembrane domain.” So in other words, they solved C2, C3, and C4 — getting the protein expressed in the right place at the right time — through some carefully completed and pretty hefty intelligently “engineered” additions of necessary sequences. These sorts of intelligently engineered macromutations do not model what nature could reasonably accomplish.
Regardless, they state: “Although the bacterial proteins function in simple assemblies, relatively little mutation would be required to convert them to function as a protein transport machine.” Is “relatively little mutation” a technical term? Spontaneously adding the proper targeting sequences, transmembrane domains, and signaling sequences is definitely not “relatively little mutation.” If anything, this research shows that evolution works best when it is intelligently “engineered.”
Rather than giving a careful account of C2-C5, the PNAS paper’s authors offer an explanation that sounds quite teleological: “Together with the LivH amino acid transporter, these component parts would have provided ‘pre-adaptation’ to bacteria ahead of a need for protein import.” Likewise, the Wired article takes care of this problem by asserting that these parts evolved, were just “ready to snap together,” and suddenly performed an entirely new function:
The process by which parts accumulate until they’re ready to snap together is called preadaptation. It’s a form of “neutral evolution,” in which the buildup of the parts provides no immediate advantage or disadvantage. Neutral evolution falls outside the descriptions of Charles Darwin. But once the pieces gather, mutation and natural selection can take care of the rest, ultimately resulting in the now-complex form of TIM23.
Wired even quotes one of the scientists who co-authored the study saying, “But when you think about it in a neutral evolutionary fashion, in which these machineries emerge before there’s a need for them, then it makes sense.”
Does it really “make sense” that the evolutionary rebuttal to irreducible complexity is sheer dumb luck, where for absolutely no apparently reason the parts for this machine just happen to be “preadapted” and “ready to snap together” and the “machineries emerge before there’s a need for them”? If this is the answer to Michael Behe, then it seems Behe’s arguments still stand.
Terms like “ready to snap together” or “pre-adaptation” sound highly goal directed, and a blind and unguided process like materialistic evolution is not supposed to be goal directed. Of course when all of these proteins are intact and functioning together, they yield a function that gives an advantage. But where did these parts suddenly come from, fully formed, in the first place, “ready to snap together” to perform this new function? When evolutionists invoke explanations like “preadaptation,” Scott Gilbert seems very right to make the critique that “[t]he modern synthesis is good at modelling the survival of the fittest, but not the arrival of the fittest.” “Preadaptation” or “ready to snap together” or “machineries emerge before there’s a need for them” are not unguided materialist explanations of evolution — like it or not they’re unwittingly appealing to a goal directed process. And there’s only one goal-directed process I know of in town: intelligent design.
These teleology-laden responses give me more confidence than ever in the potency of the challenge of irreducible complexity to evolutionary biology. It’s responses like these that made me a skeptic of neo-Darwinism and a proponent of intelligent design in the first place.
The Allegedly Simpler Machine is the Wrong Machine and Still Requires Multiple Proteins
The PNAS authors seem cognizant of the unlikelihood of these parts suddenly being “ready to snap together” and thus seek to simplify the system, asking, “could a single component of the machine function in the absence of the others to provide even inefficient protein transport?” Here they get into trouble with respect to TIM23. They’d love to find an example of just one of the TIM23 proteins functioning as a full transport mechanism somewhere, but they don’t have any evidence of that. TIM23 works in the inner membrane of the mitochondria, but there’s a similar transport mechanism on the outer membrane called TOM.
Through a genomic search, they found that in one case, TOM appears to be composed of only 1 protein complex, rather than the usual 3 or 5 — or so they say (more on this below). It’s supposed to be “proof of principle” that other transport machines could also function with only one protein:
Exhaustive analysis of the genome sequence of one group of organisms, the microsporidia, shows that they have lost the Tom22, Tom5, Tom6, and Tom7 components from their core TOM complex, and have only the Tom40 channel subunit (31).
They didn’t report any prokaryotic homologues for proteins in the TOM complex (one presumes they would have reported such had they found them), so they don’t seem to find any examples of transport mechanisms that are both potentially reducible and have prokaryotic homologues. Nonetheless, the authors make it sound like this function can be performed with only one protein in the TOM complex, but dig up their Reference 31 from the above quote, and you find a very different, and much more interesting, story.
The PNAS paper’s author’s citation for this point, Reference 31, is a January 2009 paper in the journal Eukaryotic Cell, which shows that the mitochondrial transport system in certain microsporidia (a phylum of unicellular parasites) is still very complex. (Note: microsporidia are very different from many other eukaryotes, and have a mitochondria-like organelle called the mitosome, but we’ll follow the PNAS paper’s terminology and just use the term “mitochondria” for the purposes of this response.) Look at Figure 5 on page 6 of the Eukaryotic Cell paper (which is free). On the left side in part A is the typical mitochondrial transport system known from most eukaryotes; on the right side in part B is the system for Encephalitozoon cuniculi, the species of microsporidia that allegedly has a reduced mitochondrial transport mechanism.
In Figure 5A, note that the typical eukaryotic mitochondrial transport system has redundant (i.e. 2 types of) transport machines on the inner membrane but only one type on the outer membrane. But in E. cuniculi, there’s only one transport mechanism in the inner membrane. This is the most significant extent of the “reducible complexity” that this paper reported, as seen in Figure 1 below:
Figure 1: Highly Simplified Reproduction of Figure 5 from R. F. Waller et al., “Evidence of a Reduced and Modified Mitochondrial Protein Import Apparatus in Microsporidian Mitosomes,” Eukaryotic Cell, Vol. 8(1):19-26 (Jan. 2009). Note: This diagram is highly simplified; each of the TIM and TOM complexes listed below have a number of other proteins associated with them, as well as other necessary components, in the actual diagram.
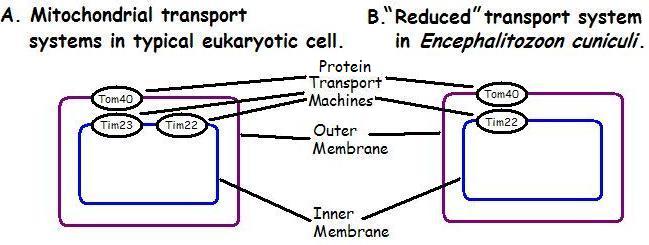
Tim23 and Tim22 have overlapping functions and redundancy to transport proteins across the inner membrane, as the Eukaryotic Cell article observes, “Mitochondrial proteins can take one of several routes to the mitochondrion via this apparatus” and complexes appear in “duplicate specialist form.” It doesn’t seem absolutely necessary to have two types of transport machines on the inner membrane if one can do the job. Is it any surprise, therefore, to find a simpler eukaryote has just one type of transport machine on the inner membrane? This isn’t a surprising finding since most higher eukaryotes seem to have redundant systems on the inner membrane.
Indeed, regarding some of the allegedly missing proteins in the E. cuniculi‘s TOM complex on the outer membrane, the paper notes that “substrate overlap between Tom20 and Tom70 occurs.” Another review of TIM and TOM complexes in the journal Current Biology likewise states:
The two [outer membrane] targeting pathways are not, however, strictly separated. Some preproteins can use both Tom70–Tom37 and Tom20–Tom22 and, most importantly, preproteins that are initially recognized by Tom70 are transferred to Tom22 before their insertion into the import pore formed by Tom40 (and possibly Tom5).
There is thus much redundancy in the typical eukaryotic transport system. But looking at Figure 5b, in the paper, note that even in the “reduced” system in E. cuniculi, at least 8 protein complexes (not one protein) are still needed to do the full job–including 4 components (not one protein) associated with the outer membrane. And contra the PNAS paper, there isn’t just one protein in the reduced TOM complex, but two.
To my knowledge, no ID proponent has ever claimed that these particular mitochondrial transport mechanisms are irreducibly complex. But what we are told is in E. cuniculi could represent an irreducible core, as it seems necessary to have one transport machine on the outer membrane and one on the inner membrane, each with a minimum number of parts, for this system to function. The Eukaryotic Cell paper confirms this point, stating:
It is possible, however, that our observations reflect an import machinery that has been reduced in response to dramatic reductionism seen throughout microsporidian biology. It is notable that, of the subset of import machinery that the HMMs have identified, all major essential functions are represented, although only once rather than in duplicate specialist form as seen in fungi and animals (Fig. 5). (emphasis added)
They’re saying that, although in higher eukaryotes there is redundancy of function (“duplicate specialist form”) for the transport systems, even in microsporidia, “all major essential functions are represented.” Thus even with a reduced yet still-complex form in E. cuniculi, it is still able to perform all the functions–a huge admission! They further state that the overall configuration shows “evidence of a conserved system, albeit in relatively minimal form”:
Our use of HMM to search E. cuniculi data shows that candidates for Tom70, Tom40, and Tim22 are identified with high confidence, and additional new components of the TIM complex (Tim50 and Pam16) and the SAM complex (Sam50) were discovered. These results offer further evidence of a conserved import system, albeit in relatively minimal form (Fig. 5).
Indeed, regarding the TOM70 protein, they state, “Despite these losses, all other major structural features identified in the yeast Tom70 structure appear to have been conserved.” (emphasis added) So it’s not reduced at all in the sense that all of the major functional components are still there in some form, and lot of proteins are necessary — at least 8 in fact — to get a protein from outside the mitochondria to the interior of the mitochondria in microsporidia.
The PNAS paper sets up a straw man claim — that the redundant mitochondrial transport system in higher eukaryotes is irreducibly complex — and then they knock it down. What we see in microsporidia may simply represent an irreducible core (see here for an explanation of the “irreducible core” concept). Try to evolve a system less complex than this, and you might not get any function.
In fact, these ID critics might be leaving off one extremely important component of this system from their irreducible core — the signal sequence on the proteins that are going to be transported. Much like a key that turns a lock, a signal on the protein is typically required for proteins to gain access into the mitochondria through these transport gates. The authors also do not consider the ability of Darwinian mechanisms to produce this lock-and-key component of the system.
Again, it must be noted that Reference 31 studies TOM, a different system than the one for which the PNAS study found homologues in bacteria (TIM23). Looking at Reference 31, we see that the simplest known mitochondrial transport system is much more complex than the PNAS paper makes it sound.
Are All E. cuniculi TOM and TIM Proteins Presently Detectable?
It’s important to note that the study in Eukaryotic Cell claimed the TOM in E. cuniculi was reduced only after a genomic study, which the investigators admit would miss proteins that were performing necessary functions but were too different from previously known TOM proteins to be detected. E. cuniculi might have more (or even all) of the standard TOM proteins fulfilling all the typical functional roles in the typical TOM system, but we wouldn’t know it from this study. Thus, the Eukaryotic Cell paper states:
The skeletal form of the mitosomal protein import machinery identified here might reflect the difficulty in identifying homologues of many of the import proteins of fungi and animals, particularly several of the small proteins. Presently few genomic data exist for microsporidia aside from E. cuniculi, limiting the opportunity to look more broadly for mitosomal homologues in this group. (emphasis added)
Again, that’s a huge admission that these allegedly reduced mitochondrial transport machines may very well be more complex than their study showed. In fact, the paper observes that “many TOM and TIM proteins show weak conservation of primary sequence,” which makes it difficult to identify them in genomic searches. In the case of some of the smaller proteins in the TOM and TIM complexes, the paper debates whether they should even expect to be able to find them:
While some of the smaller proteins (e.g., the small Toms and tiny Tims) may be difficult to recognize because of the relative simplicity of these short sequences, the absence of matches for many of the import proteins is conspicuous given that they are readily recovered from other fungal and animal genomes.
In one case they write that they aren’t sure whether the inability to identify certain proteins in these systems is due to the fact that they aren’t there, or if it’s because they are just very different from the equivalent proteins (which we’ve sequenced and are using as a basis for search) in higher eukaryotes:
It is unclear whether failure to identify further SAM complex proteins (Sam35, Sam37, and Mdm10) is due to poor conservation of these proteins or to a capacity of Sam50 to act alone.
Thus, more equivalent proteins may be present in E. cuniculi than was reported, but they may not have been detectable in their genomic search because they’re too different from presently known transport proteins. We don’t know for sure what the case is because these genomic studies don’t closely investigate the inner workings of these systems; they just scan for previously known gene sequences. If equivalent proteins are in microsporidia, then there’s no reason to believe it has a scaled down mitochondrial transport system.
Encephalitozoon cuniculi Can’t Be a Precursor to Higher Eukaryotes
Finally, it must be noted that under the prevailing phylogeny of eukaryotes, it’s impossible that the protein transport machines in the microsporidian E. cuniculi mitosomes represent some relict state before multiple TIM and TOM complexes evolved in higher eukaryotes. As the Eukaryotic Cell article states:
Given that microsporidia are most closely related to fungi and therefore diverged within the fungal-animal lineage, the ancestral microsporidia most likely possessed the common Tom and Tim proteins seen in both fungi and animals today.
Thus, the reduced transport mechanisms in E. cuniculi likely represent loss of function from the normal, more complicated state, that we find in most higher eukaryotes:
The specialization of two TIM complexes early in eukaryotic evolution enabled diverse mitochondrial protein traffic. The presence of only a single inner membrane pore in E. cuniculi suggests that microsporidia have reversed this specialization in the face of reduced protein traffic and overall cellular reduction. (emphasis added)
This organism thus cannot actually directly show some evolutionary precursor to the more advanced mitochondrial transport mechanisms we observe today.
Conclusion
The Wired article asserts that the mitochondrial transport system “seems to pose a cellular chicken-and-egg question: How could protein transport evolve when it was necessary to survive in the first place?” As can be seen, however, studies are showing that even in the “reduced” mitochondrial transport systems, “all major essential functions are represented” and many proteins are necessary to fulfill the job. The main difference is the lack of redundant systems and fewer total proteins. Wired has not solved this “chicken-and-egg question” because even the allegedly simpler system is still complex, containing multiple functional multi-protein transport machines as well as a number of required additional proteins for the transport process.
What is most revealing is that these evolutionists are forced resort to goal-directed explanatory language like “preadapted,” “parts accumulate until they’re ready to snap together,” or “machineries emerge before there’s a need for them.” Additionally, in the one case where they tried, they found it necessary to radically and intelligently “enginee[r]” a prokaryotic homologue of a transport protein to make it function in a eukaryote. No ID proponent has ever claimed these particular systems are irreducibly complex, but if these evolutionists’ arguments are any indication, then even here intelligent design seems to have the upper hand, and blind and undirected processes appear insufficient. If this paper’s explanation for the evolution of this machine, in their own words, “provides a blueprint for the evolution of cellular machinery in general,” then it’s clear that Darwinian evolutionary explanations for the origins of cellular machinery are deficient.
The PNAS article rightly states, “How these molecular machines evolved is a fundamental question.” One would think that such a fundamental question would demand a detailed, rigorous answer. Unfortunately, the PNAS authors treat this “fundamental question” as if it is solved and irreducible complexity refuted through some pretty shallow investigation and a lot of explanations that sound teleological. They want to win the debate without having one.
They are right that this is a “fundamental question,” and irreducible complexity — in the cases where ID proponents have actually argued for it — still holds much merit.
