 Evolution
Evolution
 Paleontology
Paleontology
Fossil Friday: The Abrupt Origins of Treeshrews (Scandentia) and Colugos (Dermoptera)
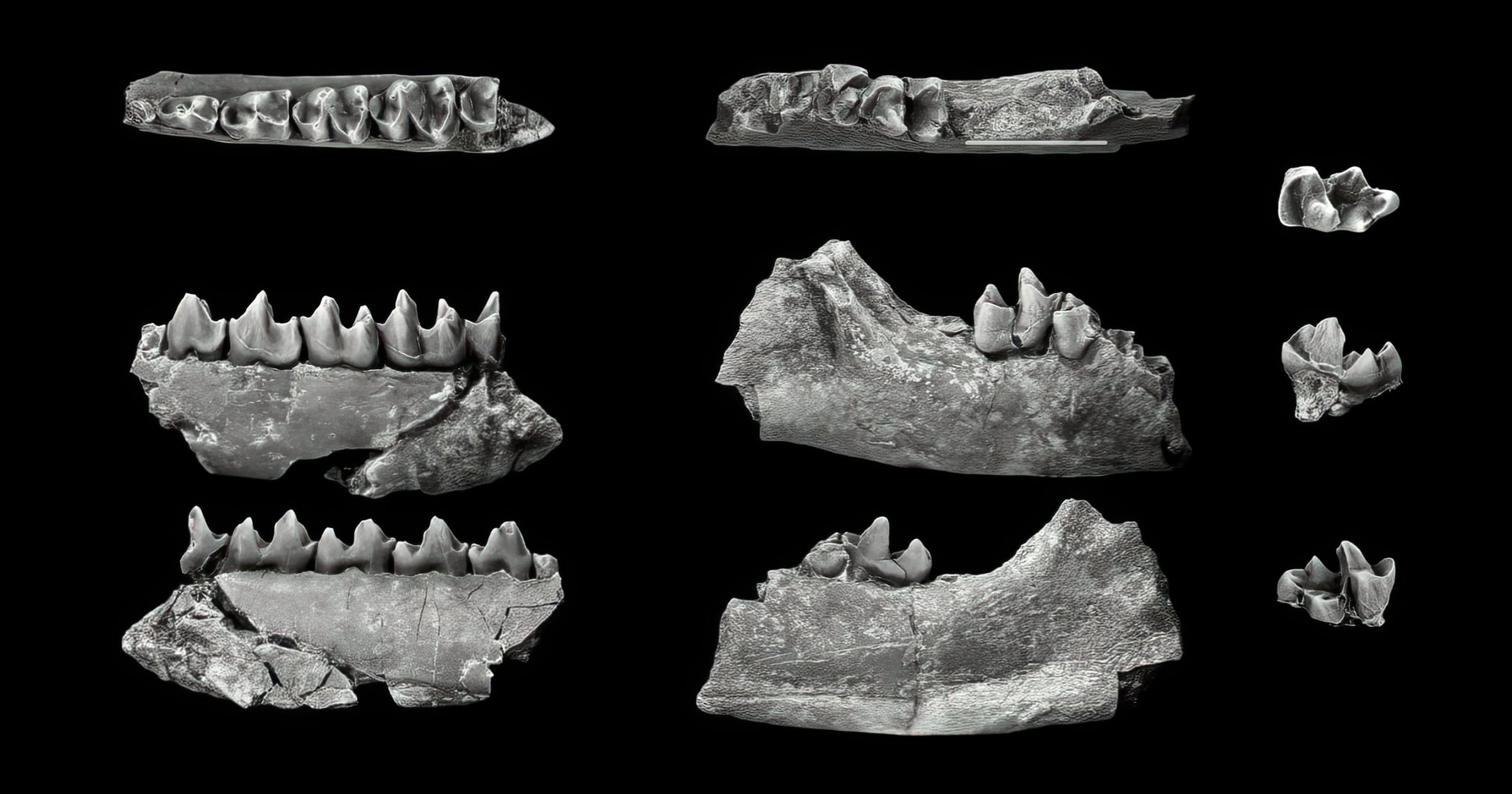
This Fossil Friday features the small mammal Eudaemonema webbi from the Late Paleocene of Western Canada (Scott 2010) as we look today into the origins of two orders of Southeast Asian placental mammals, the treeshrews (Scandentia) and colugos (Dermoptera). The 23 living species of treeshrews belong to only two families (Ptilocercidae and Tupaiidae) and look like a mixture of a shrew and a squirrel. The two living genera and species of colugos belong to the single family Cynocephalidae (= Galeopithecidae) and look like a mixture of a lemur and a flying squirrel, which is why they have sometimes been called flying lemurs. They are cat-sized arboreal animals that can glide more than 100 meters from tree to tree.
Treeshrews and colugos not only look like chimeras of different beasts, but indeed both proved to be notoriously difficult to place in the system of animals. Treeshrews were at times associated with genuine insectivores (Wagner 1855, Haeckel 1866), “menotyphlan” insectivores (esp. Macroscelidea) (Gregory 1910), and primates (Carlsson 1922, Simpson 1945, Le Gros Clark 1924, 1926, 1971, Luckett 1980, Novacek 1980, Sargis 2004). They were later removed from primates (Van Valen 1965, 1967, McKenna 1966, Szalay 1968, 1969) and recognized as a distinct order of placental mammals (Butler 1972), related to extinct Leptictida (Van Valen 1965, 1967) or of unclear relationship (Luckett 1980). This situation was even worse with colugos (Wible 1993), about which the famous German zoologist Alfred Brehm (1883) remarked in his animal encyclopedia Brehms Thierleben:
Linné stellt sie zu den Halbaffen, Cuvier zu den Fledermäusen, Geoffroy zu den Raubthieren, Oken zu den Beutelthieren und Peters endlich, wohl mit Recht, zu den Kerbthierfressern, deren Reihe sie eröffnen. Entsprechend der Unsicherheit der Forscher heißt die bekannteste Art unter anderen noch geflügelter Affe, Flattermaki, fliegende Katze, wundersame Fledermaus usw.
[Linné placed them with lemurs, Cuvier with bats, Geoffroy with carnivores, Oken with marsupials and Peters last but not least, likely correct, with insectivores right at their base. Corresponding to this uncertainty of the scientists the most common species has also been called winged monkey, flying lemur, flying cat, or wondrous bat, etc.]
Today, both orders are generally believed to be close relatives of primates in a group called Euarchonta (e.g., Sarich & Cronin 1976, Cartmill & MacPhee 1980, Adkins & Honeycutt 1991, Liu & Miyamoto 1999, Waddell et al. 1999, 2001, Liu et al. 2001, Murphy et al. 2001a, 2001b, Springer et al. 2003, 2004, 2007, Kemp 2005, Rose 2006, Kriegs et al. 2007, Halliday et al. 2015; contra Arnason et al. 2002). Together with the Glires (rodents, hares, rabbits, and pikas) the euarchontans belong to one of the four major supergroups (cohorts) of placental mammals, which has rather unimaginatively been named Euarchontoglires (Murphy et al. 2001a, 2001b) or more rarely Supraprimates (Waddell et al. 2001, Kriegs et al. 2007).
As all too often in phylogenetics the relationships of treeshrews and colugus within Euarchotoglires are still a matter of considerable scientific controversy (Knyshov et al. 2022):
Some scientists think that treeshrews are closer related to colugos in a group called Sundatheria (Adkins & Honeycutt 1991, Liu & Miyamoto 1999, Asher et al. 2009, Liu et al. 2001, Murphy et al. 2001b, Sargis 2002d, 2004, Eizirik et al. 2004, Olson et al. 2005, Marivaux et al. 2006, Bloch et al. 2002, 2007, 2016, Sánchez-Villagra et al. 2007, Nie et al. 2008, Asher & Helgen 2010, Silcox et al. 2010, 2017, Chester & Bloch 2013, O’Leary et al. 2013, Chester et al. 2015, 2017, 2019, Naish 2015, Nowak 2018, Upham et al. 2019) or Paraprimates (Springer et al. 2003, 2004, 2007). Other scientists rather believe treeshrews represent the sister group to a clade of colugos and primates called Primatomorpha (Beard 1991, 2006, Kalandadze & Rautian 1992, Murphy et al. 2001a, Waddell et al. 2001, Janečkaet al. 2007, Sargis 2007, Martin 2008, Perelman et al. 2011, Ni et al. 2013, 2016, Lin et al. 2014, Mason et al. 2016, Esselstyn et al. 2017, Boyer et al. 2018, Phillips & Fruciano 2018, Morse et al. 2019, Scornavacca et al. 2019, Zhang et al. 2019, Seiffert et al. 2020, Zachos et al. 2020, Knyshov et al. 2022, Osozawa & Wakabayashi 2023). But see Sargis (2002d, 2004), who cautioned that the support for Primatomorpha is considerably reduced when the primitive treeshrew Ptilocercus is included in the analyses. Why am I citing this boring list of all these publications? Simply to make the point very clear that the two alternative hypotheses arguably are supported by numerous independent studies based on many different data sets, but they cannot both be right. But it gets much worse.
Still other authors explicitly or implicitly suggested that treeshrews could be sister to primates only (Gregory 1910, Carlsson 1922, Le Gros Clark 1924, 1926, 1971, Simpson 1945, Simons 1964, McKenna 1966, Wible & Covert 1987, Kay et al. 1992, Novacek 1992, Kupfermann et al. 1999, Wible et al. 2007, Song et al. 2012, Kumar et al. 2013, Lin et al. 2014, Zhou et al. 2015), even though some of these studies did not include colugos in their analyses. A few scientists suggested treeshrews as the sister group of Glires+Primatomorpha together (Kumar et al. 2013, Esselstyn et al. 2017, Knyshov et al. 2022), or even suggested them to be the sister group of Glires (Meredith et al. 2011, Zhou et al. 2015, Foley et al. 2016; but see Lin et al. 2014), or of Lagomorpha (Bailey et al. 1992), or of Rodentia (Arnason et al. 2002), which would all imply that Euarchonta and Sundatheria would not be valid clades (also see Madsen et al. 2001). Finally, some studies recovered Dermoptera nested within primates as sister group of Anthropoidea (Murphy et al. 2001a, Arnason et al. 2002, contra Schmitz et al. 2002 and Schmitz & Zischler 2003).
Originally, scientists believed that the order of bats (Chiroptera) belongs to the same supergroup as colugos and primates (Archonta sensu lato) (e.g., Gregory 1910, McKenna 1975, Wible & Covert 1987, Novacek 1992, Szalay & Lucas 1993, Kupfermann et al. 1999). Prior to the advent of molecular phylogenetics most scientists believed that bats and colugos belong to a common clade that was named Volitantia (Szalay & Drawhorn 1980, Novacek & Wyss 1986, Wible & Covert 1987, Wible & Novacek 1988, Baker et al. 1991, Novacek 1992, Simmons 1993, 1995, Szalay & Lucas 1993, 1996, Wible 1993, Stafford & Thorington 1998, Bloch & Silcox 2001, Silcox 2001a, Sargis 2002d, 2002e, 2007, Silcox et al. 2005; also see Halliday et al. 2015: fig. 1). This was based on a substantial number of anatomical similarities, mainly related to gliding/flying adaptations, but also including the morphology of the teeth and the ear capsule. Based on features of penis morphology Smith & Madkour (1980) suggested a clade of only Dermoptera + Megachiroptera as sister group of primates, with tree shrews and Microchiroptera as more basal outgroups. The results from modern phylogenomics did not agree at all and consequently bats were ultimately removed from archontans (Asher & Helgen 2010) and are now considered as basal members of completely different supergroup called Laurasiatheria. Prior to this recognition there were some wild theories seriously discussed, such as the diphyly of bats and the “fallen angel” hypothesis (Pettigrew et al. 1989; contra Bailey et al. 1992), which suggested that primates derived from a gliding common ancestor with colugos and megabats. Nothing seems impossible or forbidden in Darwinian fantasy land, except anything that smacks of purposeful development and design.
So, let’s focus on the hard evidence, and what could be harder than petrified fossils? Unfortunately, the fossil record of treeshrews and colugos is quite sparse, but it still provides some useful information about their origins. This evidence strongly contradicts the Darwinian predictions from molecular clock studies, which suggested that colugos should have branched from the Primatomorpha lineage about 79.6 million years ago, and treeshrews even earlier about 86.2 million years ago during the Cretaceous “golden age” of dinosaurs (Janečka et al. 2007). Roberts et al. (2011: fig. 3) accordingly suggested that the two families of crown group treeshrews already diverged in the Paleocene about 60 million years ago. Foley et al. (2016) proposed a similar estimate with the lineages of treeshrews originating 76.94 million years ago and colugos 75.47 million years ago. Of course, the fossils tell a very different story that is better agreeing with recalibrated datings of a “soft explosive model of placental mammal evolution” (Phillips & Fruciano 2018, also see Upham et al. 2019: fig. 4), which has all the orders appearing abruptly during a brief window of time in the early Paleogene. This is exactly what we heretical ID proponents always said.
The Fossil Record of Treeshrews
It is worth noting that not only the affinity of treeshrews and the intraordinal relationships among the living species of treeshrews proved to be a contentious issue (Olson et al. 2004, 2005, Roberts et al. 2011), but even the very number of species itself, which for example varied in the genus Tupaia between 11 and 32 species (Olson et al. 2005). The fossil record of treeshrews is relatively poor (Sargis 1999, 2004, Olson et al. 2004, 2005, Rose 2006). The oldest fossil record of the order Scandentia is Eodendrogale parvum that was described by Tong (1988), based on a few isolated teeth from the Middle Eocene (48.6-37.2 mya) of Xichuan in China. A few extinct members of modern treeshrews have been described from Miocene localities in East Asia, such as Prodendrogale and Tupaia storchi from the Late Miocene (11.1-4.9 mya) of Yunnan in China, and Palaeotupaia and Sivatupaia from the Miocene and Pliocene (23.03-5.33 mya) Śiwalik deposits in India and Pakistan (Dutta 1975, Chopra & Vasishat 1979, Chopra et al. 1979, Jacobs 1980, Qiu 1986, Sargis 1999, 2004, Ni & Qiu 2012, Sehgal et al. 2022), as well as Tupaia miocenica from the Miocene (ca. 18 mya) of Thailand (Mein & Ginsburg 1997). The oldest crown group treeshrew is Ptliocercus kylinfrom the Earliest Oligocene (ca. 34 mya) of the Yunnan Province in China (Li & Ni 2016), which has been interpreted as evidence that treeshrews are slowly evolving “living fossils.” The press release about the discovery also mentioned that morphological comparisons and phylogenetic analysis support the long-standing idea that the pen-tailed treeshrews of the living relict species Ptilocercus lowii “are morphologically conservative and have probably retained many characters present in the common stock that gave rise to archontans, which include primates, flying lemurs, plesiadapiforms and treeshrews” (Chinese Academy of Sciences 2016; see also Sargis 2002a, 2002b, 2002c, 2002d, 2007 and Olson et al. 2005). That seems to be a rather bold conclusion considering the above-mentioned fact that scientists cannot even agree on the phylogenetic affinities of treeshrews in the first place.
Some fossil taxa that were previously assigned to the relationship of treeshrews have meanwhile been debunked: Following Lemoine (1885), the extinct Adapisoriculidae were considered as fossil Tupaiidae by Simpson (1928), Van Valen (1965, 1967) and Szalay (1968). Most later studies rather considered them to be lipothylan insectivores (e.g., Rose 2006), but Smith et al. (2010) made a strong case for a position among basal Euarchonta. However, more recent studies even disputed their position within crown group placental mammals (Goswami et al. 2011, Manz et al. 2015). Matthew (1918) provisionally listed the Eocene genus Entomolestes as a possible fossil Tupaiidae, but it was later recognized as a close relative of erinaceoid insectivores (= hedgehogs) by McKenna (1966) and Novacek et al. (1985). McKenna (1966) also considered other suggested candidates as very doubtful, such as the genera Macrocranion (likely a hedgehog as well) and the Paleocene mixodectid Eudaemonema that we featured in this article. The latter genus was considered as a Plagiomenidae within Dermoptera by KcKenna (1960) and McKenna & Bell (1997), which was arguably corroborated by the cladistic analysis of Ni et al. (2013, 2016 SI). The Paleogene family Anagalidae was considered as closely related to Tupaiidae by Simpson (1931), but this was strongly disputed by McKenna (1966). Anagalids are today considered as closer related to Glires within a clade Gliriformes, which we will look into more detail next week’s Fossil Friday.
The Fossil Record of Colugos
The two species of the extinct genus Dermotherium from the Eocene and Oligocene of Thailand, Myanmar, and Pakistan are the oldest and only definitive fossil dermopterans (Ducrocq et al. 1992, Marivaux et al. 2006). The older of these two species is Dermotherium major from the Late Eocene (37.2-33.9 mya) of the Krabi Basin in Thailand. Stafford & Szalay (2000) cautioned that this purported dermopteran fossil is poorly preserved and of little help, but the affinity to modern Dermoptera was corroborated by Silcox et al. (2005) and Smith et al. (2010). Apparently they were already quite similar to modern colugos and were therefore included in the same family Cynocephalidae together with the two living genera, which are by the way much more distinct than was often believed (Stafford & Szalay 2000). Some alleged fossil dermopterans have been reported from Neogene localities in Africa (see PaleoDB), but these seem to be only brief records in obscure checklists, which have been totally ignored in the technical literature on dermopteran evolution. Several enigmatic Paleogene groups of small insectivorous mammals have also been associated with Dermoptera (Anonymous 2023) and merit a closer look.
Plagiomenidae
This extinct family is known exclusively from the Paleocene and Early Eocene of North America. According to Bloch et al. (2007) they belong to Sundatheria, together with colugos and treeshrews. Several authors had more specifically attributed this family to Dermoptera (Matthew 1918, Simpson 1937, 1945, Romer 1966, Van Valen 1967, Szalay 1969, Rose 1973, 1975, 2006, Krishtalka & Setoguchi 1977, Rose & Simons 1977, Bown & Rose 1979, Novacek 1980, Carroll 1988, Gunnell 1989, McKenna 1990, Ducrocq et al. 1992, McKenna & Bell 1997, Silcox 2001a, 2001b, Agusti & Antón 2002, Kemp 2005). Such a position was also confirmed by the cladistic studies of Ni et al. (2013, 2016 SI), Halliday et al. (2015), and Morse et al. (2019). On the other hand, MacPhee et al. (1989) considered plagiomenids in his seminal study as eutherians of uncertain affinity, which was concurred by Marivaux et al. (2006). Szalay& Lucas (1993) also cautioned that the affinity of Plagiomenidae needs reexamination, and Dawson et al. (1993) remarked:
The phylogenetic position of Plagiomenidae with respect to other mammals is also not yet clear. Earlier interpretations of plagiomenids as members of the order Dermoptera have been questioned on several grounds (MacPhee et al. 1989, Beard 1990, Kay et al. 1990). At present, we follow MacPhee et al. (1989) in classifying these animals as placental mammals of unknown ordinal affinities.
A lot of this uncertainty concerning the affinities of Plagiomenidae, and the other taxa of fossil small mammals mentioned below, comes from the fact that the fragmentary fossil evidence is mostly restricted to dental characters. Yapuncich et al. (2011) reported the first dentally associated skeleton of Plagiomenidae, which surprisingly did not exhibit any arboreal adaptations, so that the authors concluded that “on functional morphological and cladistic grounds we consider Plagiomenidae to be more likely allied with laurasiatheres than dermopterans or other euarchontans.” Oopsy, there goes almost a hundred years of previous research down the drain. In this context, it is interesting that more recent studies of living treeshrews suggested that the arboreal adaptations already belonged to the archontan ground plan and thus do not suggest a uniquely primate relationship of treeshrews (Godinot 2017).
Anyway, What About the Age of Plagiomenids?
Three species in the genus Plagiomene and Planetetherium mirabile are known from Late Paleocene and Early Eocene (56.8-50.3 mya) localities in North America (Matthew 1918, Simpson 1928). The genus Thylacaelurus, which was described from the Middle Eocene Kishenehn Formation in Canada (Russell 1954), has also been reported from the Paleocene (61.7-56.8 mya) Paskapoo Formation in Alberta, Canada (Fox 1990), but only in a list without any description, figure, or justification. McKenna (1990) included three more genera (Tarka, Tarkadectes, and Ekgmowechashala) from the Middle Eocene and Oligocene of northwestern USA, all classified in a separate plagiomenid subfamily Ekgmowechashalinae. This subfamily was recently recognized as a family of adapiform primates by Ni et al. (2016). Another genus and species Ellesmene eureka has been described from Early Eocene (55.8-50.3 mya) of the Arctic region of Ellesmere Island (Dawson et al. 1993), which had a subtropical climate and vegetation during this period of earth history, but still a polar light regime that made it to a very unique environment that was also colonized by other early Primatomorpha like the plesiadapiform genus Ignacius (Miller et al. 2023).
There are two more taxa that may belong to Plagiomenidae:
Worlandia inusitata was described from the Paleocene (Clarkforkian, 56.8-55.8 mya) of Wyoming and considered to be closely related to plagiomenids like Planetetherium in a subfamily Worlandiinae (Bown & Rose 1979). This was accepted by McKenna & Bell (1997)and Rose (2006), and the cladistic study of Paleocene mammals by Halliday et al. (2015)supported its place within Plagiomenidae.
The genus Elpidophorus was described by Simpson (1927, 1937) with two species from the Paleocene (61.7-56.8 mya) of Montana, Wyoming, and Alberta (Fox 1990). It was originally described by Simpson (1927) as a carnivoran, but was attributed to Mixodectidae by most early workers (Simpson 1936, 1937, 1945, Van Valen 1967, Szalay 1969). McKenna (1960) begged to differ and considered Elpidopherus as a plesiadapiform stem primate. This genus was later transferred from Mixodectidae to Plagiomenidae and considered as earliest putative dermopteran by Rose (1975). This was concurred by several subsequent studies (Gunnell 1989, Fox 1990, McKenna & Bell 1997, and Halliday et al. 2015), while Scott et al. (2013) again treated this genus as Mixodectidae. Ni et al. (2013 SI) placed it again with Plagiomenidae in the stem group of Dermoptera, far removed from Mixodectes. So, it looks like Elpidophorus could be the oldest plagiomenid (Rose 2006) or not, related to dermopterans or not.
Mixodectidae
The Mixodectidae represent another extinct family from the Paleocene of North America and are almost exclusively known from their dentition (Simpson 1937, Szalay 1969, Gunnell 1989, Rose 2006, 2008). They have been linked previously with rodents, insectivores (Gunnel 1989), and primates (see McKenna 1966, Szalay 1969, Silcox 2001a: fig. 6.4, and Scott 2010), as well as attributed to (eu)archontans with an affinity to Plagiomenidae and Dermoptera (Simpson 1937, Van Valen 1967, Carroll 1988, Beard 1989, McKenna 1990, Szalay & Lucas 1993, 1996, Silcox et al. 2005, Gunnell & Silcox 2008, Rose 2006, 2008, Scott 2010). Szalay (1968, 1969) already reviewed the checkered history of the taxonomic allocation of mixodectids. He rejected a close relationship with plagiomenids and dermopterans and instead considered mixodectids and adapisoriculids as close relatives of treeshrews. MacPhee et al. (1989) affirmed a sister group relationship of Mixodectidae with Plagiomenidae (also see Rose & Simons 1977, McKenna 1990, Rose 2008), but considered them as Eutheria incertae sedis. Agusti & Antón (2002) considered mixodectids as “archaic placental mammals”. Some more recent studies indeed rather considered mixodectids to belong to the plesiadapiform stem group of Primatomorpha (Ni et al. 2013, 2016 SI) than that of Dermoptera, but Ni et al. (2013, 2016) placed the putative mixodectid Eudaemonema not together with Mixodectes but in the stem group of Dermoptera. Some dental similarities of Mixodectidae with recent colugos have been interpreted as convergences (Scott 2010). Well, that does not help much.
Micromomyidae
Micromomyidae was an extinct family of diminutive euarchontan mammals that lived from the Late Paleocene to the Early Eocene of western North America (with a questionable record from the Eocene of China; Tong & Wang 2006). Because postcranial material was interpreted in terms of an adaptation to gliding behaviour, this family has also been linked with Dermoptera (e.g., Beard 1989, 1993a, 1993b). However, this interpretation as mitten-gliders was arguably refuted by Bloch et al. (2007) and Boyer & Bloch (2008). More recently micromomyids were rather assigned to the plesiadapiform grade in the stem group of primates (Silcox 2001a, Silcox et al. 2005, 2010, 2017, Rose 2006, Bloch et al. 2007, Chester & Bloch 2013, Chester et al. 2015, 2017, 2019, Bloch et al. 2016) or of Primatomorpha (Ni et al. 2013 SI). The cranial inflation shared with dermopterans could more likely be a convergence (Bloch et al. 2016).
Microsyopidae
This extinct family also existed in the Paleocene and Eocene of North America. It has been suggested as member of the stem group of Sundatheria (treeshrews and colugos) by the cladistic study of Bloch & Silcox (2006), but recovered as stem dermopterans by Ni et al. (2013, 2016 SI). Beard (1989) also placed them with plesiadapiforms and Dermoptera. Szalay & Lucas (1993) affirmed an inclusion in Archonta but remained undecided about the specific affinities. However, more recent cladistic studies recovered this family as plesiadapiform-grade stem primates (Silcox et al. 2005, 2010, 2017, Chester & Bloch 2013, Chester et al. 2015, 2017, 2019, Bloch et al. 2016). Indeed, the majority of experts had long considered microsyopids as close relatives of primates or even included them as basal primates (McKenna 1960, 1966, Van Valen 1967, Szalay 1969, MacPhee & Cartmill 1986, Gunnell 1989, Silcox 2001a, and Rose 2006).
Plesiadapiformes: To Be or Not to Be a Glider
As we already discussed in my Fossil Friday article on the origin of primates (Bechly 2022), some scientists considered the Paleogene mammal order of Plesiadapiformes as possible close relatives of colugos (Dermoptera) (Kemp 2005, Silcox 2014, Godinot 2017). This was mainly based on the shared reduction of the internal carotid artery (Kay et al. 1990, 1992) and some skeletal characters that were thought to be indicative of an adaptation to gliding in paromomyid genera like Phenacolemur and Ignacius (Beard 1989, 1990, 1993a, 1993b, Martin 1990, McKenna & Bell 1997). Szalay & Lucas (1993) found homologies in the postcranial skeleton. As plesiadapiforms include some of the oldest known placental mammals at all (see Bechly 2022), this could be a very remarkable finding concerning the early origin of the colugo lineage. However, this proposed relationship was seriously questioned by many other experts (Krause 1991, Ducrocq et al. 1992, Szalay & Lucas 1993, 1996, Wible 1993, Van Valen 1994, Stafford & Szalay 2000, Boyer et al. 2001, Bloch & Silcox 2001, 2006, Sargis 2002d, Bloch & Boyer 2002a, 2002b, 2003, Silcox 2001a, 2001b, 2003, Rose 2006, Bloch et al. 2007, and Boyer & Bloch 2008). Simons (1964) had already cautioned that the similarities between Plesiadapis and colugos “could have been acquired independently rather than from a common ancestor.” Also, the cladistic studies by Bloch et al. (2007, 2016), Janečka et al. (2007), and Chester et al. (2015, 2017, 2019), found no evidence supporting a dermopteran relationship of plesiadapiforms and instead recovered them as basal grade in the stem group of primates. But phylogenetics would not be phylogenetics if there would not be an even more recent and more comprehensive cladistic analysis that again confirmed the close relationship of some plesiadapiforms (incl. the putative primate Altiatlasius, also see Ni et al. 2016 SI) and colugos (Morse et al. 2019), and even Boyer et al. (2018: fig. 9) again clustered some plesiadapiforms (incl. the type genus Plesiadapis) with Dermoptera. Sigh, what a frustrating mess indeed!
Last but not least, there is an extinct family Placentidentidae with the single genus and species Placentidens lotus from the Early Eocene (Ypresian, 55.8-48.6 mya) of France, which was attributed to Dermoptera by some scientists (Russell et al. 1973, Carroll 1988, Ducrocq et al. 1992). Rose & Simons (1977) considered Placentidens as a possible Plagiomenidae and thus dermopteran too. However, this genus was more recently shown to belong to the extinct family Nyctitheriidae in the insectivore suborder Soricomorpha (Beard & Dawson 2009), thus related to the true shrews and moles in the totally different supergroup Laurasiatheria. Well, unless you follow Smith et al. (2010), who said that “the purported euarchontan Paleogene family Nyctitheriidae (Hooker 2001) is closer to Scandentia than to adapisoriculids.” But wait, Manz et al. (2015) again found that Nyctitheriidae is related neither to Euarchonta nor to Adapisoriculidae, but to Eulipotyphla, thus true insectivores. Is there anything the experts can agree upon beyond trivial facts like those beasts being extinct small mammals? They all look at the same fossil evidence and constantly come to totally different conclusions. Even as a paleontologist I have to admit that calling this a real scientific discipline seems like an insult to hard sciences like physics or chemistry or molecular biology. To an outsider it must rather resemble a kind of Rorschach test with fossils instead of ink blotches, and all that matters seems to be guesswork, speculation, and opinion.
Long story short: Irrespective of any of the numerous uncertainties, treeshrews and colugos definitely appeared abruptly in the Paleogene. The fossil record shows nothing even remotely resembling a gradual origin of these orders in the Cretaceous that was predicted by Darwinian molecular clock studies. This is just another instance of the countless empirical failures of the theory, more or less ignored by mainstream evolutionary biology.
Next Fossil Friday we will look into the origins of the orders of rodents and Lagomorpha, which form the second major clade (Glires) within the supergroup of Euarchontoglires. I hope that will not be as confusing and wearying as today’s topic.
References
- Adkins RM & Honeycutt RL 1991. Molecular phylogeny of the superorder Archonta. PNAS88(22), 10317–10321. DOI: https://doi.org/10.1073/pnas.88.22.10317
- Agusti J & Antón M 2002. Mammoths, Sabertooths, and Hominids: 65 Million Years of Mammalian Evolution in Europe. Columbia University Press, New York (NY), 328 pp.
- Anonymous 2023. Colugos: The fossil record. Gliding Mammals of the World. https://www.myym.ru/colugos/1.html
- Arnason U, Adegoke JA, Bodin K, Born EW, Esa YB, Gullberg A, Nilsson M, Short RV, Xu X & Janke A 2002. Mammalian mitogenomic relationships and the root of the eutherian tree. PNAS 99(12), 8151–8156. DOI: https://doi.org/10.1073/pnas.102164299
- Asher RJ & Helgen KM 2010. Nomenclature and placental mammal phylogeny. BMC Evolutionary Biology 10:102, 1–9. DOI: https://doi.org/10.1186/1471-2148-10-102
- Asher RJ, Bennett N & Lehmann T 2009. The new framework for understanding placental mammal evolution. BioEssays 31(8), 853–864. DOI: https://doi.org/10.1002/bies.200900053
- Bailey WJ, Slightom JL & Goodman M 1992. Rejection of the “Flying Primate” Hypothesis by Phylogenetic Evidence from the ε-globin Gene. Science 256(5053), 86–89. DOI: https://doi.org/10.1126/science.1301735
- Baker RJ, Novacek MJ & Simmons NB 1991. On the monophyly of bats. Systematic Zoology 40(2), 216–231. DOI: https://doi.org/10.1093/sysbio/40.2.216
- Beard KC 1989. Postcranial anatomy, locomotor adaptations, and paleoecology of early Cenozoic Plesiadapidae, Paromomyidae, and Micromomyidae (Eutheria, Dermoptera). PhD Dissertation, The Johns Hopkins University, 1322 pp.
- Beard KC 1990. Gliding behaviour and palaeoecology of the alleged primate family Paromomyidae (Mammalia, Dermoptera). Nature 345(6273), 340–341. DOI: https://doi.org/10.1038/345340a0
- Beard KC 1991. Vertical postures and climbing in the morphotype of Primatomorpha: Implications for locomotor evolution in primate history. pp. 79–87 in: Coppens Y & Senut B (eds). Origine(s) de la Bipédie chez les Hominidés. Cahiers de Paleoanthropologie. Editions du CNRS, Paris (F), 338 pp.
- Beard KC 1993a. Phylogenetic systematics of the Primatomorpha, with special reference to Dermoptera. pp. 129–150 in: Szalay FS, Novacek MJ & McKenna MC (eds.). Mammal Phylogeny: Placentals. Springer-Verlag, Berlin (DE), xi+321 pp.
- Beard KC 1993b. Origin and Evolution of Gliding in Early Cenozoic Dermoptera (Mammalia, Primatomorpha). pp. 63–90 in: MacPhee RDE (ed.). Primates and Their Relatives in Phylogenetic Perspective. Springer, New York (NY), xiv+384 pp. DOI: https://doi.org/10.1007/978-1-4899-2388-2_2
- Beard KC 2006. Mammalian Biogeography and Anthropoid Origins. pp. 439–467 in: Lehman SM & Fleagle JG (eds). Primate Biogeography: Progress and Prospects. Springer, New York (NY), xii+536 pp. DOI: https://doi.org/10.1007/0-387-31710-4_15
- Beard KC & Dawson MR 2009. Early Wasatchian Mammals of the Red Hot Local Fauna, Uppermost Tuscahoma Formation, Lauderdale County, Mississippi. Annals of Carnegie Museum 78(3), 193–243. DOI: https://doi.org/10.2992/007.078.0301
- Bechly G 2022. Fossil Friday: Purgatorius and the Abrupt Origin of Primates. Evolution News December 9, 2022. https://evolutionnews.org/2022/12/fossil-friday-purgatorius-and-the-abrupt-origin-of-primates/
- Bloch JI & Boyer DM 2002a. Grasping primate origins. Science 298(5598), 1606–1610. DOI: https://doi.org/10.1126/science.1078249
- Bloch JI & Boyer DM 2002b. Phalangeal morphology of Paleocene Plesiadapiformes (Mammalia: ?Primates): evaluation of the gliding hypothesis and the first evidence for grasping in a possible primate ancestor. The Geological Society of America Abstracts with Programs 34, 13.
- Bloch JI & Boyer DM 2003. Response to comment on “Grasping primate origins”. Science300(5620), 741c. DOI: https://doi.org/10.1126/science.1082060
- Bloch JI & Silcox MT 2001. New basicrania of Paleocene–Eocene Ignacius: Re-evaluation of the Plesiadapiform-Dermopteran link. American Journal of Physical Anthropology 116(3), 184–198. DOI: https://doi.org/10.1002/ajpa.1114
- Bloch JI & Silcox MT 2006. Cranial anatomy of Paleocene plesiadapiform Carpolestes simpsoni (Mammalia, Primates) using ultra high-resolution X-ray computed tomography, and the relationships of plesiadapiforms to Euprimates. Journal of Human Evolution 50(1), 1–35. DOI: https://doi.org/10.1016/j.jhevol.2005.06.009
- Bloch JI, Silcox MT & Sargis EM 2002. Origin and relationships of Archonta (Mammalia, Eutheria): re-evaluation of Eudermoptera and Primatomorpha. Journal of Vertebrate Paleontology 22(S3), 37A.
- Bloch JI, Silcox MT, Boyer DM & Sargis EJ 2007. New Paleocene skeletons and the relationship of plesiadapiformes to crown-clade primates. PNAS 104(4), 1159–1164. DOI: https://doi.org/10.1073/pnas.0610579104
- Bloch JI, Chester SGB & Silcox MT 2016. Cranial anatomy of Paleogene Micromomyidae and implications for early primate evolution. Journal of Human Evolution 96, 58–81. DOI: https://doi.org/10.1016/j.jhevol.2016.04.001
- Bown TM & Rose KD 1979. Mimoperadectes, A New Marsupial, and Worlandia, A New Dermopteran, from the Lower Part of the Willwood Formation (Early Eocene), Bighorn Basin, Wyoming. Contributions from the Museum of Paleontology, University of Michigan25(4), 89–104. https://hdl.handle.net/2027.42/48494
- Boyer DM & Bloch JI 2008. Evaluating the Mitten-Gliding Hypothesis for Paromomyidae and Micromomyidae (Mammalia, “Plesiadapiformes”) Using Comparative Functional Morphology of New Paleogene Skeletons. Chapter 11, pp. 233–284 in: Sargis EJ & Dagosto M (eds). Mammalian Evolutionary Morphology: A Tribute to Frederick S. Szalay. Springer, Dordrecht (NL), xxviii+439 pp.DOI: https://doi.org/10.1007/978-1-4020-6997-0_11
- Boyer DM, Bloch JI & Gingerich PD 2001. New skeletons of Paleocene paromomyids (Mammalia, ?Primates): were they mitten gliders? Journal of Vertebrate Paleontology21(S3), 35A. DOI: https://doi.org/10.1080/02724634.2001.10010852
- Boyer DM, Maiolino SA, Holroyd PA, Morse PE & Bloch JI 2018. Oldest evidence for grooming claws in euprimates. Journal of Human Evolution 122, 1–22. DOI: https://doi.org/10.1016/j.jhevol.2018.03.010
- Brehm AE 1883 (1876–1879, reprinted 1882–1884). Brehms Tierleben. Allgemeine Kunde des Tierreichs. 2nd expanded edition in 10 volumes. Bibliographisches Institut, Leipzig (DE). [In German]
- Butler PM 1972. The problem of insectivore classification. pp. 253-265 in: Joycey KA & Kemp TS (eds). Studies in Vertebrate Evolution. Oliver & Boyd, Edinburgh (UK), ix+284 pp.
- Carlsson A 1922. Über die Tupaiidae und ihre Beziehungen zu den Insectivora und den Prosimiae. Acta Zoologica 3(2-3), 227–270. DOI: https://doi.org/10.1111/j.1463-6395.1922.tb01021.x
- Carroll RL 1988. Vertebrate Paleontology and Evolution. WH Freeman & Co, New York (NY), xiv+698 pp.
- Cartmill M & MacPhee RDE 1980. Tupaiid affinities: the evidence of the carotid arteries and cranial skeleton. pp. 95–132 in: Luckett WP (ed.). Comparative Biology and Evolutionary Relationships of Tree Shrew. Springer, New York (NY), xv+314 pp. DOI: https://doi.org/10.1007/978-1-4684-1051-8_3
- Chester SGB & Bloch JI 2013. Systematics of Paleogene Micromomyidae (Euarchonta, Primates) from North America. Journal of Human Evolution 65(2), 109 –142. DOI: https://doi.org/10.1016/j.jhevol.2013.04.006
- Chester SGB, Bloch JI, Boyer DM & Clemens WA 2015. Oldest known euarchontan tarsals and affinities of Paleocene Purgatorius to Primates. PNAS 112(5), 1487–1492. DOI: https://doi.org/10.1073/pnas.1421707112
- Chester SGB, Williamson TE, Bloch JI, Silcox MT, Sargis EJ. 2017 Oldest skeleton of a plesiadapiform provides additional evidence for an exclusively arboreal radiation of stem primates in the Palaeocene. Royal Society Open Science 4(5):170329, 1–9. DOI: https://doi.org/10.1098/rsos.170329
- Chester SGB, Williamson TE, Silcox MT, Bloch JI & Sargis EJ 2019. Skeletal morphology of the early Paleocene plesiadapiform Torrejonia wilsoni (Euarchonta, Palaechthonidae). Journal of Human Evolution 128, 76–92. DOI: https://doi.org/10.1016/j.jhevol.2018.12.004
- Chinese Academy of Sciences 2016. Earliest-known treeshrew fossil found in Yunnan, China. Phys.org January 26, 2016. https://phys.org/news/2016-01-earliest-known-treeshrew-fossil-yunnan-china.html
- Chopra SRK & Vasishat RN 1979. Śivalik fossil tree shrew from Haritalyangar, India. Nature281(5728), 214–215. DOI: https://doi.org/10.1038/281214a0
- Chopra SRK, Kaul S & Vasishat RN 1979. Miocene tree shrews from the Indian Sivaliks. Nature 281(5728), 213–214. DOI: https://doi.org/10.1038/281213a0
- Dawson MR, McKenna MC, Beard KC & Hutchinson JH 1993. An Early Eocene Plagiomenid Mammal from Ellesmere and Axel Heiberg Islands, Arctic Canada. Kaupia 3, 179–192. https://www.researchgate.net/publication/237067091
- Ducrocq S, Buffetaut E, Buffetaut-Tong H, Jaeger J-J, Jongkanjanasoontorn Y & Suteethorn V 1992. First fossil flying lemur: a dermopteran from the Late Eocene of Thailand. Palaeontology 35(2), 373–380. https://www.palass.org/publications/palaeontology-journal/archive/35/2/article_pp373-380
- Dutta AK 1975. Micromammals from Siwaliks. Indian Minerals 29, 76–77.
- Eizirik E, Murphy WJ, Springer MS, O’Brien SJ 2004. Molecular Phylogeny and Dating of Early Primate Divergences. pp. 45–64 in: Ross CF & Kay RF (eds). Anthropoid Origins – New Visions. Springer, New York (NY), xxix+749 pp. DOI: https://doi.org/10.1007/978-1-4419-8873-7_2
- Esselstyn JA, Oliveros CH, Swanson MT & Faircloth BC 2017. Investigating Difficult Nodes in the Placental Mammal Tree with Expanded Taxon Sampling and Thousands of Ultraconserved Elements. Genome Biology and Evolution 9(9), 2308–2321. DOI: https://doi.org/10.1093/gbe/evx168
- Foley NM, Springer MS & Teeling EC 2016. Mammal madness: Is the mammal tree of life not yet resolved?. Philosophical Transactions of the Royal Society B 371(1699):20150140, 1-11. DOI: https://doi.org/10.1098/rstb.2015.0140
- Fox RC 1990. The succession of Paleocene mammals in western Canada. pp. 51–70 in: Bown TM & Rose KD (eds). Dawn of the Age of Mammals in the Northern Part of the Rocky Mountain Interior, North America. GSA Special Papers 243. DOI: https://doi.org/10.1130/SPE243-p51
- Godinot M 2017. Paleocene and Eocene Primates. pp. 1–9 in: Fuentes A (ed.). The International Encyclopedia of Primatology. John Wiley & Sons, Hoboken (NJ), 1608 pp. DOI: https://doi.org/10.1002/9781119179313.wbprim0331
- Goswami A, Prasad GVR, Upchurch P, Boyer DM, Seiffert ER, Verma O, Gheerbrant E & Flynn JJ 2011. A radiation of arboreal basal eutherian mammals beginning in the late Cretaceous of India. PNAS 108(39), 16333–16338. DOI: https://doi.org/10.1073/pnas.1108723108
- Gregory WK 1910. The orders of mammals. Bulletin of the American Museum of Natural History 27, 1–524. DOI: http://hdl.handle.net/2246/313
- Gunnell GF 1989. Evolutionary History of Microsyopoidea (Mammalia, ?Primates) and the Relationship Between Plesiadapiformes and Primates. University of Michigan Papers on Paleontology 27, viii+157 pp. https://lsa.umich.edu/paleontology/publications/papers-on-paleontology/27-The-Evolutionary-History-of-Microsyopoidea-Mammalia-Primates-and-the-Relationship-between-Plesiadapiformes-and-Primates.html
- Gunnel GF & Silcox MT 2008. Archonta summary. pp. 161–173 in: Janis CM, Scott KM & Jacobs LL (eds). Evolution of Tertiary Mammals of North America. Vol. 2: Small Mammals, Xenarthrans, and Marine Mammals. Cambridge University Press, Cambridge (MA), 802 pp.
- Haeckel E 1866. Generelle Morphologie der Organismen. Georg Reimer, Berlin (DE), 574 pp.
- Halliday TJD, Upchurch P & Goswami A 2015. Resolving the relationships of Paleocene placental mammals. Biological Reviews 92(1), 521–550. DOI: https://doi.org/10.1111/brv.12242
- Hooker JJ 2001. Tarsals of the extinct insectivoran family Nyctitheriidae (Mammalia): evidence for archontan relationships. Zoological Journal of the Linnean Society 132(4), 501–529. DOI: https://doi.org/10.1111/j.1096-3642.2001.tb02473.x
- Jacobs LL 1980. Siwalik fossil tree shrews. pp. 205–216 in: Luckett WP (ed.). Comparative Biology and Evolutionary Relationships of Tree Shrews. Plenum Press, New York (NY), xv+314 pp.
- Janečka JE, Miller W, Pringle TH, Wiens F, Zitzmann A, Helgen KM, Springer MS & Murphy WJ 2007. Molecular and genomic data identify the closest living relative of primates. Science 318(5851), 792–794. DOI: https://doi.org/10.1126/science.1147555
- Kalandadze NN & Rautian SA 1992. Systema mlekopitayushchikh i istorygeskaya zoogeographei [The system of mammals and historical zoogeography]. Sbornik Trudov Zoologicheskogo Muzeya Moskovskogo Goschdarstvennoro Universiteta 29, 44–152 [In Russian].
- Kay RF, Thorington Jr RW & Houde P 1990. Eocene plesiadapiform shows affinities with flying lemurs not primates. Nature 345(6273), 342–344. DOI: https://doi.org/10.1038/345342a0
- Kay RF, Thewissen JGM & Yoder AD 1992. Cranial anatomy of Ignacius graybullianus and the affinities of the Plesiadapiformes. American Journal of Physical Anthropology 89(4), 477–498. DOI: https://doi.org/10.1002/ajpa.1330890409
- Kemp TS 2005. The Origin and Evolution of Mammals. Oxford University Press, Oxford (UK), 331 pp. [Google Books]
- Knyshov A, Hyrtsenko Y, Literman R & Schwartz RS 2022. Interrogating Genomic Data in the Phylogenetic Placement of Treeshrews Reveals Potential Sources of Conflict. bioRxivpreprint. DOI: https://doi.org/10.1101/2021.11.18.469131
- Krause DW 1991. Were paromomyids gliders? Maybe, maybe not. Journal of Human Evolution 21(3), 177–188. DOI: https://doi.org/10.1016/0047-2484(91)90060-9
- Kriegs JO, Churakov G, Jurka J, Brosius J & Schmitz J 2007. Evolutionary history of 7SL RNA-derived SINEs in Supraprimates. Trends in Genetics 23(4): 158–161. DOI: https://doi.org/10.1016/j.tig.2007.02.002
- Kumar V, Hallström BM & Janke A 2013. Coalescent-Based Genome Analyses Resolve the Early Branches of the Euarchontoglires. PLoS ONE 8(4):e60019, 1–9. DOI: https://doi.org/10.1371/journal.pone.0060019
- Kupfermann H, Satta Y, Takahata N, Tichy H & Klein J 1999. Evolution of Mhc–DRB Introns: Implications for the Origin of Primates. Journal of Molecular Evolution 48(6), 663–674. DOI: https://doi.org/10.1007/pl00006510
- Le Gros Clark WE 1924. On the Brain of the Tree-Shrew (Tupaia minor). Proceedings of the Zoological Society of London 94(4), 1053–1074. DOI: https://doi.org/10.1111/j.1096-3642.1924.tb03328.x
- Le Gros Clark WE 1926. On the Anatomy of the Pen-tailed Tree-Shrew(Ptilocercus lowii). Proceedings of the Zoological Society of London 96(4), 1179–1309. DOI: https://doi.org/10.1111/j.1096-3642.1926.tb02241.x
- Le Gros Clark WE 1971. The Antecedents of Man: An Introduction to the Evolution of the Primates. 3rd edition. Edinburgh University Press, Edinburgh (UK), 374 pp.
- Lemoine V 1885. Étude sur quelques Mammifères de petite taille de la faune cernaysienne des environs de Reims. Bulletin de la Société Geologique de France 13, 203–217. https://www.biodiversitylibrary.org/item/181692#page/238/mode/1up
- Li Q & Ni X 2016. An early Oligocene fossil demonstrates treeshrews are slowly evolving “living fossils”. Scientific Reports 6(1):18627, 1–8. DOI: https://doi.org/10.1038/srep18627
- Lin J, Chen G, Gu L, Shen Y, Zheng M, Zheng W, Hu X, Zhang X, Qiu Y, Liu X & Jiang C 2014. Phylogenetic affinity of tree shrews to Glires is attributed to fast evolution rate. Molecular Phylogenetics and Evolution 71, 193–200. DOI: https://doi.org/10.1016/j.ympev.2013.12.001
- Liu F-GR & Miyamoto MM 1999. Phylogenetic assessment of molecular and morphological data for eutherian mammals. Systematic Biology 48(1), 54–64. DOI: https://doi.org/10.1080/106351599260436
- Liu F-GR, Miyamoto MM, Freire NP, Ong PQ, Tennant MR, Young TS & Gugel KE 2001. Molecular and Morphological Supertrees for Eutherian (Placental) Mammals. Science291(5509), 1786–1789. DOI: https://doi.org/10.1126/science.1056346
- Luckett WP 1980. The suggested evolutionary relationships and classification of tree shrews. pp. 3–31 in: Luckett WP (ed.). Comparative Biology and Evolutionary Relationships of Tree Shrews. Plenum Press, New York (NY), xv+314 pp. DOI: https://doi.org/10.1007/978-1-4684-1051-8_1
- MacPhee RDE & Cartmill M 1986. Basicranial structures and primate systematics. pp. 219–275 in Swindler DR & Erwin J (eds). Comparative Primate Biology. Vol. 1: Systematics, Evolution, and Anatomy. Alan R. Liss, New York (NY), xvi+836 pp.
- MacPhee RDE, Cartmill M & Rose KD 1989. Craniodental morphology and relationships of the supposed Eocene dermopteran Plagiomene (Mammalia). Journal of Vertebrate Paleontology 9(3), 329–349. DOI: https://doi.org/10.1080/02724634.1989.10011766
- Madsen O, Scally M, Douady CJ, Kao DJ, DeBry RW, Adkins R, Amrine HM, Stanhope MJ, Jong WW & Springer MS 2001. Parallel adaptive radiations in two major clades of placental mammals. Nature 409(6820), 610–614. DOI: https://doi.org/10.1038/35054544
- Manz CL, Chester SGB, Bloch JI, Silcox MT & Sargis EJ 2015. New partial skeletons of Palaeocene Nyctitheriidae and evaluation of proposed euarchontan affinities. Biology Letters 11:20140911, 1–5. DOI: https://doi.org/10.1098/rsbl.2014.0911
- Marivaux L, Bocat L, Chaimanee Y, Jaeger J-J, Marandat B, Srisuk P, Tafforeau P, Yarmee C & Welcomme J-L 2006. Cynocephalid dermopterans from the Palaeogene of South Asia (Thailand, Myanmar and Pakistan): systematic, evolutionary and palaeobiogeographic implications. Zoologica Scripta 35(4), 395–420. DOI: https://doi.org/10.1111/j.1463-6409.2006.00235.x
- Martin RD. 1990. Some relatives take a dive. Nature 345(6273), 291–292. DOI: https://doi.org/10.1038/345291a0
- Martin RD 2008. Colugos: obscure mammals glide into the evolutionary limelight. Journal of Biology 7(4):13, 1–5. DOI: https://doi.org/10.1186/jbiol74
- Mason VC, Li G, Minx P, Schmitz J, Churakov G, Doronina L, Melin AD, Dominy NJ, Lim NT-L, Springer MS, Wilson RK, Warren WC, Helgen KM & Murphy WJ 2016. Genomic analysis reveals hidden biodiversity within colugos, the sister group to primates. Science Advances 2(8):e1600633, 1–15. DOI: https://doi.org/10.1126/sciadv.1600633
- Matthew WD 1918. A revision of the Lower Eocene Wasatch and Wind River faunas. Part V. Insectivora (Continued), Glires, Edentata. Bulletin of the American Museum of Natural History 38(16), 565–657. http://hdl.handle.net/2246/1352
- McKenna MC 1960. Fossil Mammalia from the early Wasatchian Four Mile fauna, Eocene of northwest Colorado. University of California Publications in Geological Sciences 37(1), 1–130.
- McKenna MC 1966. Paleontology and the origin of Primates. Folia Primatologica 4(1), 1–25. DOI: https://doi.org/10.1159/000155041
- McKenna MC 1975. Toward a Phylogenetic Classification of the Mammalia. pp. 21–46 in: Luckett WP & Szalay FS (eds.). Phylogeny of the primates: a multidisciplinary approach. Proceedings of WennerGren Symposium no. 61, Burg Wartenstein, Austria, July 6–14, 1974. Plenum Press, New York (NY). DOI: https://doi.org/10.1007/978-1-4684-2166-8_2
- McKenna MC 1990. Plagiomenids (Mammalia: ?Dermoptera) from the Oligocene of Oregon, Montana, and South Dakota, and middle Eocene of northwestern Wyoming. pp. 211–234 in: Bown TM & Rose KD (eds). Dawn of the Age of Mammals in the Northern Part of the Rocky Mountain Interior, North America. GSA Special Papers 243.DOI: https://doi.org/10.1130/SPE243-p211
- McKenna MC & Bell SK 1997. Classification of Mammals Above the Species Level. Colombia University Press, New York (NY), xii+631 pp. https://books.google.at/books?id=zS7FZkzIw-cC
- Mein P & Ginsburg L 1997. Les mammifères du gisement miocène inférieur de Li Mae Long, Thaïlande: systématique, biostratigraphie et paléoenvironnement. Géodiversitas19(4), 783–844. https://biostor.org/reference/243911
- Meredith RW, Janečka JE, Gatesy J et al. 2011. Impacts of the Cretaceous terrestrial revolution and KPg extinction on mammal diversification. Science 334(6055), 521–524. DOI: https://doi.org/10.1126/science.1211028
- Miller K, Tietjen K & Beard KC 2023. Basal Primatomorpha colonized Ellesmere Island (Arctic Canada) during the hyperthermal conditions of the early Eocene climatic optimum. PLoS ONE. https://doi.org/10.1371/journal.pone.0280114
- Morse PE, Chester SGB, Boyer DM, Smith T, Smith R, Gigase P & Bloch JI 2019. New fossils, systematics, and biogeography of the oldest known crown primate Teilhardina from the earliest Eocene of Asia, Europe, and North America. Journal of Human Evolution 128, 103–131. DOI: https://doi.org/10.1016/j.jhevol.2018.08.005
- Murphy WJ, Eizirik E, Johnson WE, Zhang YP, Ryder OA & O’Brien SJ 2001a. Molecular phylogenetics and the origins of placental mammals. Nature 409(6820), 614–618. DOI: https://doi.org/10.1038/35054550
- Murphy WJ, Eizirik E, O’Brien SJ, Madsen O, Scally M, Douady CJ, Teeling E, Ryder OA, Stanhope MJ, de Jong WW & Springer MS 2001b. Resolution of the Early Placental Mammal Radiation using Bayesian phylogenetics. Science 294(5550), 2348–2351. DOI: https://doi.org/10.1126/science.1067179
- Naish D 2015. Introducing the Treeshrews: They Don’t All Live in Trees and They Aren’t Close to Shrews. Scientific American blogs Tetrapod Zoology December 4, 2015. https://blogs.scientificamerican.com/tetrapod-zoology/introducing-the-treeshrews/
- Ni X & Qiu Z 2012 Tupaiine tree shrews (Scandentia, Mammalia) from the Yuanmou Lufengpithecus locality of Yunnan, China. Swiss Journal of Palaeontology 131(1), 51–60. DOI: https://doi.org/10.1007/s13358-011-0029-0
- Ni X, Gebo DL, Dagosto M, Meng J, Tafforeau P, Flynn JJ & Beard KC 2013. The oldest known primate skeleton and early haplorhine evolution. Nature 498(7452), 60–64 and Supplementary Information. DOI: https://doi.org/10.1038/nature12200
- Ni X, Li Q, Li L & Beard KC 2016. Oligocene primates from China reveal divergence between African and Asian primate evolution. Science 352(6286), 673–677 and Supplementary Information. DOI: https://doi.org/10.1126/science.aaf2107
- Nie W, Fu B, O’Brien PCM, Wang J, Su W, Tanomtong A, Volobouev V, Ferguson-Smith MA & Yang F 2008. Flying lemurs – the ’flying tree shrews’? Molecular cytogenetic evidence for a Scandentia-Dermoptera sister clade. BMC Biology 6:18, 1–11. DOI: https://doi.org/10.1186/1741-7007-6-18
- Novacek MJ 1980. Cranioskeletal features in tupaiids and selected Eutheria as phylogenetic evidence. pp. 35–93 in: Luckett WP (ed.). Comparative Biology and Evolutionary Relationships of Tree Shrew. Springer, New York (NY), xv+314 pp. DOI: https://doi.org/10.1007/978-1-4684-1051-8_2
- Novacek MJ 1992. Mammalian phylogeny: shaking the tree. Nature 356(6365), 121–125. DOI: https://doi.org/10.1038/356121a0
- Novacek MJ & Wyss AR 1986. Higher-level relationships of recent eutherian orders: morphological evidence. Cladistics 2(4), 257–287. DOI: https://doi.org/10.1111/j.1096-0031.1986.tb00463.x
- Novacek MJ, Bown TM & Schankler D 1985. On the classification of early Tertiary Erinaceomorpha (Insectivora, Mammalia). American Museum Novitates 2813, 1–22.http://hdl.handle.net/2246/5283
- Nowak RM 2018. Walker’s Mammals of the World: Monotremes, Marsupials, Afrotherians, Xenarthrans, and Sundatherians. Johns Hopkins University Press, Baltimore (MD), 784 pp.
- O’Leary MA, Bloch JI, Flynn JJ et al. 2013. The Placental Mammal Ancestor and the Post–K-Pg Radiation of Placentals. Science 339 (6120), 662–667. DOI: https://doi.org/10.1126/science.1229237
- Olson LE, Sargis EJ & Martin RD 2004. Phylogenetic relationships among treeshrews (Scandentia): a review and critique of the morphological evidence. Journal of Mammalian Evolution 11, 49–71.DOI: https://doi.org/10.1023/B:JOMM.0000029145.28207.6d
- Olson LE, Sargis EJ & Martin RD 2005. Intraordinal phylogenetics of treeshrews (Mammalia: Scandentia) based on evidence from the mitochondrial 12S rRNA gene. Molecular Phylogenetics and Evolution 35(3), 656–673. DOI: https://doi.org/10.1016/j.ympev.2005.01.005
- Osozawa S & Wakabayashi J 2023. Geologically calibrated mammalian tree and the corresponding global events, including birth of human. Research Square preprint, 33 pp. DOI: https://doi.org/10.21203/rs.3.rs-523676/v1
- Perelman P, Johnson WE, Roos C et al. 2011. A Molecular Phylogeny of Living Primates. PLoS Genetics 7(3):e1001342, 1–17. DOI: https://doi.org/10.1371/journal.pgen.1001342
- Pettigrew JD, Jamieson BGM, Robson SK, Hall LS, McAnally KI & Cooper HM 1989. Phylogenetic relations between microbats, megabats and primates (Mammalia: Chiroptera and primates). Philosophical Transactions of the Royal Society B 325(1229), 489–559. DOI: https://doi.org/10.1098/rstb.1989.0102
- Phillips MJ & Fruciano C 2018. The soft explosive model of placental mammal evolution. BMC Evolutionary Biology 18:104, 1–13. DOI: https://doi.org/10.1186/s12862-018-1218-x
- Qiu Z 1986. Fossil tupaiid from the hominoid locality of Lufeng, Yunnan. Vertebrata PalAsiatica 24(4), 308–319. http://www.ivpp.cas.cn/cbw/gjzdwxb/xbwzxz/200903/P020100315566703286848.pdf
- Roberts TE, Lanier HC, Sargis EJ & Olson LE 2011. Molecular phylogeny of treeshrews (Mammalia: Scandentia) and the timescale of diversification in Southeast Asia. Molecular Phylogenetics and Evolution 60(3), 358–372. DOI: https://doi.org/10.1016/j.ympev.2011.04.021
- Romer AS 1966. Vertebrate Paleontology. 3rd edition. University of Chicago Press, Chicago (IL), 468 pp.
- Rose KD 1973. The mandibular dentition of Plagiomene (Dermoptera, Plagiomenidae). Breviora 411, 1–17.https://biostor.org/reference/4130
- Rose KD 1975. Elpidophorus, the Earliest Dermopteran (Dermoptera, Plagiomenidae). Journal of Mammology 56(3), 676–679. DOI: https://doi.org/10.2307/1379482
- Rose KD 2006. The Beginning of the Age of Mammals. John Hopkins University Press, Baltimore (MA), xiv+428 pp. https://books.google.at/books?id=3bs0D5ix4VAC
- Rose KD 2008. Plagiomenidae and Mixodectidae. Chapter 13, pp. 198-206 in: Janis CM, Gunnell GF & Uhen MD (eds). Evolution of Tertiary Mammals of North America. Volume 2: Small Mammals, Xenarthrans, and Marine Mammals. Cambridge University Press, Cambridge (MA), viii+795 pp. DOI: https://doi.org/10.1017/CBO9780511541438.014
- Rose KD & Simons EL 1977. Dental Function in the Plagiomenidae: Origin and Relationships of the Mammalian Order Dermoptera. Contributions from the Museum of Paleontology, University of Michigan 24(20), 221–236. https://hdl.handle.net/2027.42/48487
- Russell LS 1954. Mammalian fauna of the Kishenehn Formation, southeastern British Columbia. Canada National Museum Bulletin 132, 92–111.
- Russell DE, Louis P & Savage DE 1973. Chiroptera and Dermoptera of the French Early Eocene. University of California Publications in Geological Sciences 95, 1–57.
- Sánchez‐Villagra MR, Narita Y & Kuratani S 2007. Thoracolumbar vertebral number: The first skeletal synapomorphy for afrotherian mammals. Systematics and Biodiversity 5(1), 1–7. DOI: https://doi.org/10.1017/s1477200006002258
- Sargis EJ 1999. Tree shrews. pp. 1286–1287 in: Singer R (ed.). Encyclopedia of Paleontology. Fitzroy Dearborn, Chicago (IL), 1550 pp.
- Sargis EJ 2002a. Functional morphology of the forelimb of tupaiids (Mammalia, Scandentia) and its phylogenetic implications. Journal of Morphology 253(1), 10–42. DOI: https://doi.org/10.1002/jmor.1110
- Sargis EJ 2002b. Functional morphology of the hindlimb of tupaiids (Mammalia, Scandentia) and its phylogenetic implications. Journal of Morphology 254(2), 149–185. DOI: https://doi.org/10.1002/jmor.10025
- Sargis EJ 2002c. A multivariate analysis of the postcranium of tree shrews (Scandentia, Tupaiidae) and its taxonomic implications. Mammalia 66(4), 579–598. DOI: https://doi.org/10.1515/mamm.2002.66.4.579
- Sargis EJ 2002d. The postcranial morphology of Ptilocercus lowii (Scandentia, Tupaiidae): an analysis of primatomorphan and volitantian characters. Journal of Mammalian Evolution9(1/2), 137–160. DOI: https://doi.org/10.1023/A:1021387928854
- Sargis EJ 2002e. Primate origins nailed. Science 298(5598), 1564–1565. DOI: https://doi.org/10.1126/science.1079034
- Sargis EJ 2004. New views on tree shrews: the role of Tupaiids in primate supraordinal relationships. Evolutionary Anthropology 13(2), 56–66. DOI: https://doi.org/10.1002/evan.10131
- Sargis EJ 2007. The Postcranial Morphology of Ptilocercus lowii (Scandentia, Tupaiidae) and its Implications for Primate Supraordinal Relationships. pp. 51–82 in: Ravosa MJ & Dagosto M (eds). Primate Origins: Adaptations and Evolution. Springer, New York (NY), xxx+829 pp. DOI: https://doi.org/10.1007/978-0-387-33507-0_3
- Sarich VM & Cronin JE 1976. Molecular systematics of the primates. pp. 141–170 in: Goodman M, Tashian RE & Tashian JH (eds). Molecular Anthropology. Plenum Press, New York (NY) xiii+466 pp. DOI: https://doi.org/10.1007/978-1-4615-8783-5_8
- Schmitz J & Zischler H 2003. A novel family of tRNA-derived SINEs in the colugo and two new retrotransposable markers separating dermopterans from primates. Molecular Phylogenetics and Evolution 28(2), 341–349. DOI: https://doi.org/10.1016/s1055-7903(03)00060-5
- Schmitz J, Ohme M, Suryobroto B & Zischler H 2002. The Colugo (Cynocephalus variegatus, Dermoptera): The Primates’ Gliding Sister? Molecular Biology and Evolution19(12), 2308–2312. DOI: https://doi.org/10.1093/oxfordjournals.molbev.a004054
- Scornavacca C, Belkhir K, Lopez J, Dernat R, Delsuc F, Douzery EJP & Ranwez V 2019. OrthoMaM v10: Scaling–up orthologous coding sequence and exon alignments with more than one hundred mammalian genomes. Molecular Biology and Evolution 36(4), 861–862. DOI: https://doi.org/10.1093/molbev/msz015
- Scott CS 2010. Eudaemonema webbi sp. nov. (Mammalia, Mixodectidae) from the late Paleocene of western Canada: the youngest known mixodectid. Canadian Journal of Earth Sciences 47(12), 1451–1462. DOI: https://doi.org/10.1139/e10-074
- Scott CS, Spivak DN & Sweet AR 2013. First mammals from the Paleocene Porcupine Hills Formation of southwestern Alberta, Canada. Canadian Journal of Earth Sciences 50(3), 355–378. DOI: https://doi.org/10.1139/e2012-044
- Sehgal RK, Singh AP, Gilbert CC, Patel BA, Campisano CJ, Selig KR, Patnaik R & Singh NP 2022. A new genus of treeshrew and other micromammals from the middle Miocene hominoid locality of Ramnagar, Udhampur District, Jammu and Kashmir, India. Journal of Paleontology 96(6), 1318–1335. DOI: https://doi.org/10.1017/jpa.2022.41
- Seiffert ER, Tejedor MF, Fleagle JG, Novo NM, Cornejo FM, Bond M, de Vries D & Campbell jr KE 2020. A parapithecid stem anthropoid of African origin in the Paleogene of South America. Science 368(6487), 194–197. DOI: https://doi.org/10.1126/science.aba1135
- Silcox MT 2001a. A phylogenetic analysis of Plesiadapiformes and their relationship to Euprimates and other archontans. Ph.D. Dissertation, Johns Hopkins University, School of Medicine, Baltimore (MD), xviii+729 pp.
- Silcox MT 2001b. A phylogenetic analysis of Plesiadapiformes and their relationship to Euprimates and other archontans. Journal of Vertebrate Paleontology, 101A.
- Silcox MT 2003. New discoveries on the middle ear anatomy of Ignacius graybullianus(Paromomyidae, Primates) from ultra high resolution X-ray computed tomography. Journal of Human Evolution 44(1), 73–86. DOI: https://doi.org/10.1016/S0047-2484(02)0195-1
- Silcox MT 2014. Primate Origins and the Plesiadapiforms. Nature Education Knowledge 5(3), 1. https://www.nature.com/scitable/knowledge/library/primate-origins-and-the-plesiadapiforms-106236783/
- Silcox MT, Bloch JI, Boyer DM, Sargis EJ 2005. Euarchonta (Dermoptera, Scandentia, Primates). Chapter 9, pp. 127–144 in: Rose KD & Archibald JD (eds). The Rise of Placental Mammals: Origins and Relationships of the Major Extant Clades. Johns Hopkins University Press, Baltimore (MD), 280 pp. https://www.researchgate.net/profile/Jonathan-Bloch-3/publication/259104117_Euarchonta/links/0c960529fae0318cce000000/Euarchonta.pdf
- Silcox MT, Bloch JI, Boyer DM & Houde P 2010. Cranial anatomy of Paleocene and Eocene Labidolemur kayi (Mammalia: Apatotheria), and the relationships of the Apatemyidae to other mammals. Zoological Journal of the Linnean Society 160(4), 773–825. DOI: https://doi.org/10.1111/j.1096-3642.2009.00614.x
- Silcox MT, Bloch JI, Boyer DM, Chester SGB & López-Torres S 2017. The evolutionary radiation of plesiadapiforms. Evolutionary Anthropology 26(2), 74–94. DOI: https://doi.org/10.1002/evan.21526
- Simmons NB 1993. The Importance of Methods: Archontan Phylogeny and Cladistic Analysis of Morphological Data. pp. 1–61 in: MacPhee RDE (ed.). Primates and Their Relatives in Phylogenetic Perspective. Plenum Press, New York (NY), xiv+384 pp. DOI: https://doi.org/10.1007/978-1-4899-2388-2_1
- Simmons NB 1995. Bat relationships and the origin of flight. Symposia of the Zoological Society of London 67, 27–43.
- Simons EL 1964. The Early Relatives of Man. Scientific American 211(1), 50–65. https://www.jstor.org/stable/24931561
- Simpson GG 1927. Mammalian fauna and correlation of the Paskapoo Formation of Alberta. American Museum Novitates 268, 1–10. DOI: http://hdl.handle.net/2246/3144
- Simpson GG 1928. A new mammalian fauna from the Fort Union of southern Montana. American Museum Novitates 297, 1–15. http://hdl.handle.net/2246/3165
- Simpson GG 1931. A new insectivore from the Oligocene, Ulan Gochu horizon, of Mongolia. American Museum Novitates 505, 1–22. http://hdl.handle.net/2246/2983
- Simpson GG 1936. A new fauna from the Fort Union of Montana. American Museum Novitates 873, 1–27. http://hdl.handle.net/2246/2164
- Simpson GG 1937. The Fort Union of the Crazy Mountain field, Montana and its mammalian faunas. Bulletin of the United States National Museum 169, x+287 pp. DOI: https://doi.org/10.5479/si.03629236.169.1
- Simpson GG 1945. The Principles of Classification and a Classification of the Mammals. Bulletin American Museum of Natural History 85, ix+350 pp. http://hdl.handle.net/2246/1104
- Smith JD & Madkour G 1980. Penial morphology and the question of chiropteran phylogeny. pp. 347–365 in: Wilson DE & Gardner AL (eds). Proceedings of the Fifth International Bat Research Conference. Texas Tech Press, Lubbock (TX), 434 pp.
- Smith T, De Bast E & Sigé B 2010. Euarchontan affinity of Paleocene Afro-European adapisoriculid mammals and their origin in the late Cretaceous Deccan Traps of India. Naturwissenschaften 97, 417–422.DOI: https://doi.org/10.1007/s00114-010-0651-5
- Song S, Liu L, Edwards SV & Wu S 2012. Resolving conflict in eutherian mammal phylogeny using phylogenomics and the multispecies coalescent model. PNAS 109(37), 14942–14947. DOI: https://doi.org/10.1073/pnas.1211733109
- Springer MS, Murphy WJ, Eizirik E & O’Brien SJ 2003. Placental mammal diversification and the Cretaceous–Tertiary boundary. PNAS 100(3), 1056–1061. DOI: https://doi.org/10.1073/pnas.0334222100
- Springer MS, Stanhope MJ, Madsen O & de Jong WW 2004. Molecules consolidate the placental mammal tree. Trends in Ecology and Evolution 19(8), 430–438. DOI: https://doi.org/10.1016/j.tree.2004.05.006
- Springer MS, Murphy WJ, Eizirik E, Madsen O, Scally M, Douady CJ, Teeling EC, Stanhope MJ, de Jong WW & O’Brien SJ 2007. A molecular classification for the living orders of placental mammals of the phylogenetic placement of primates. Chapter 1, pp. 1–28 in: Ravosa MJ & Dagosto M (eds). Primate Origins: Adaptations and Evolution. Springer, New York (NY), xxx+829 pp. DOI: https://doi.org/10.1007/978-0-387-33507-0_1
- Stafford BJ & Szalay FS 2000. Craniodental functional morphology and taxonomy of dermopterans. Journal of Mammalogy 81(2), 360–385. DOI: https://doi.org/10.1644/1545-1542(2000)081<0360:CFMATO>2.0.CO;2
- Stafford BJ & Thorington RW 1998. Carpal development and morphology in archontan mammals. Journal of Morphology 235(2), 135–155. DOI: https://doi.org/10.1002/(SICI)1097-4687(199802)235:2<135::AID-JMOR4>3.0.CO;2-8
- Szalay FS 1968. The beginnings of primates. Evolution 22(1), 19–36. DOI: https://doi.org/10.1111/j.1558-5646.1968.tb03445.x
- Szalay FS 1969. Mixodectidae, Microsyopidae and the insectivore–primate transition. Bulletin of the American Museum of Natural History 140:4, 193–330. http://hdl.handle.net/2246/1130
- Szalay FS & Drawhorn G 1980. Evolution and Diversification of the Archonta in an Arboreal Milieu. pp. 133–169 in: Luckett PW (ed.). Comparative Biology and Evolutionary Relationships of Tree Shrews. Plenum Press, New York (NY), xv+314 pp. DOI: https://doi.org/10.1007/978-1-4684-1051-8_4
- Szalay FS & Lucas SG 1993. Cranioskeletal morphology of archontans, and diagnoses of Chiroptera, Volitantia, and Archonta. pp. 187–226 in: MacPhee RDE (ed.). Primates and their Relatives in Phylogenetic Perspective. Springer, New York (NY), xiv+384 pp. DOI: https://doi.org/10.1007/978-1-4899-2388-2_6
- Szalay FS & Lucas SG 1996. The postcranial morphology of Paleocene Chriacus and Mixodectes and the phylogenetic relationships of archontan mammals. New Mexico Museum of Natural History & Science Bulletin 7, 1–47. https://nmdigital.unm.edu/digital/collection/bulletins/id/7126/
- Tong Y 1988. Fossil tree shrews from the Eocene Hetaoyuan Formation of Xichuan, Henan. Vertebrata PalAsiatica 26(3), 214–220. http://www.ivpp.cas.cn/cbw/gjzdwxb/xbwzxz/200902/W020090813373091049969.pdf
- Tong Y & Wang J 2006. Fossil Mammals from the Early Eocene Wutu Formation of Shandong Province. Palaeontologica Sinica NS C 192(28), 1–195 [In Chinese with English summary].
- Upham NS, Esselstyn JA & Jetz W 2019. Inferring the mammal tree: Species-level sets of phylogenies for questions in ecology, evolution, and conservation. PLoS Biology17(12):e3000494, 1–44. DOI: https://doi.org/10.1371/journal.pbio.3000494
- Van Valen L 1965. Treeshrews, primates and fossils. Evolution 19(2), 137–151. https://www.jstor.org/stable/2406368
- Van Valen L 1967. New Paleocene insectivores and insectivore classification. Bulletin of the American Museum of Natural History 135(5), 217–284. http://hdl.handle.net/2246/358
- Van Valen L 1994. The origin of the plesiadapid primates and the nature of Purgatorius. Evolutionary Monographs 15, 1–79.
- Waddell PJ, Okada N & Hasegawa M 1999. Towards Resolving the Interordinal Relationships of Placental Mammals. Systematic Biology 48(1), 1–5. DOI: https://doi.org/10.1093/sysbio/48.1.1
- Waddell PJ, Kishino H & Ota R 2001. A Phylogenetic Foundation for Comparative Mammalian Genomics. Genome Informatics 12, 141–154. https://pubmed.ncbi.nlm.nih.gov/11791233/
- Wagner JA 1855. Die Säugethiere in Abbildungen nach der Natur. Supplementband, Abt. 5. Weiger, Leipzig (DE), 810 pp.
- Wible JR 1993. Cranial circulation and relationships of the colugo Cynocephalus(Dermoptera, Mammalia). American Museum Novitates 3072, 1–27. http://hdl.handle.net/2246/4969
- Wible JR & Covert HH 1987. Primates: cladistic diagnosis and relationships. Journal of Human Evolution 16(1), 1–22.DOI: https://doi.org/10.1016/0047-2484(87)90058-3
- Wible JR & Novacek MJ 1988. Cranial evidence for the monophyletic origin of bats. American Museum Novitates 2911, 1–19. http://hdl.handle.net/2246/5168
- Wible JR, Rougier GW, Novacek MJ & Asher RJ 2007. Cretaceous eutherians and Laurasian origin for placental mammals near the K/T boundary. Nature 447(7147), 1003–1006. DOI: https://doi.org/10.1038/nature05854
- Yapuncich GS, Boyer DM, Secord R & Bloch JI 2011. The first dentally associated skeleton of Plagiomenidae (Mammalia, ?Dermoptera) from the late Paleocene of Wyoming. Abstracts of the 71st Annual Meeting of the Society of Vertebrate Paleontology, Las Vegas (NV), November 2011, 218. DOI: https://doi.org/10.13140/2.1.1302.4322
- Zachos FE 2020. Mammalian Phylogenetics: A Short Overview of Recent Advances. pp. 1–18 in: Hackländer K & Zachos F (eds). Mammals of Europe – Past, Present, and Future. Handbook of the Mammals of Europe. Springer, Cham (CH), 130 pp. DOI: https://doi.org/10.1007/978-3-319-65038-8_6-1
- Zhang M-L, Li M-L, Ayoola AO, Murphy RW, Wu D-D & Shao Y 2019. Conserved sequences identify the closest living relatives of primates. Zoological Research 40(6), 532–540. DOI: https://doi.org/10.24272/j.issn.2095-8137.2019.057
- Zhou X, Sun F, Xu S, Yang G & Li M 2015. The position of tree shrews in the mammalian tree: Comparing multi-gene analyses with phylogenomic results leaves monophyly of Euarchonta doubtful. Integrative Zoology 10(2), 186–198. DOI: https://doi.org/10.1111/1749-4877.12116
