 Evolution
Evolution
 Intelligent Design
Intelligent Design
The Mystery of Energy Metabolism

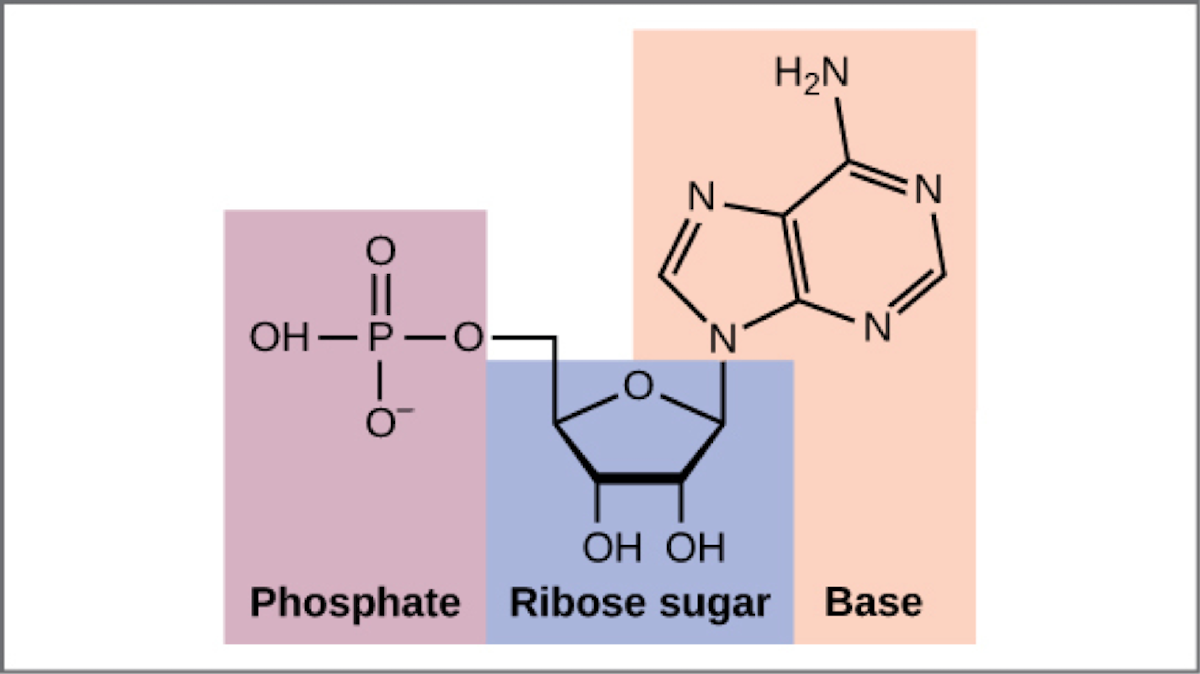
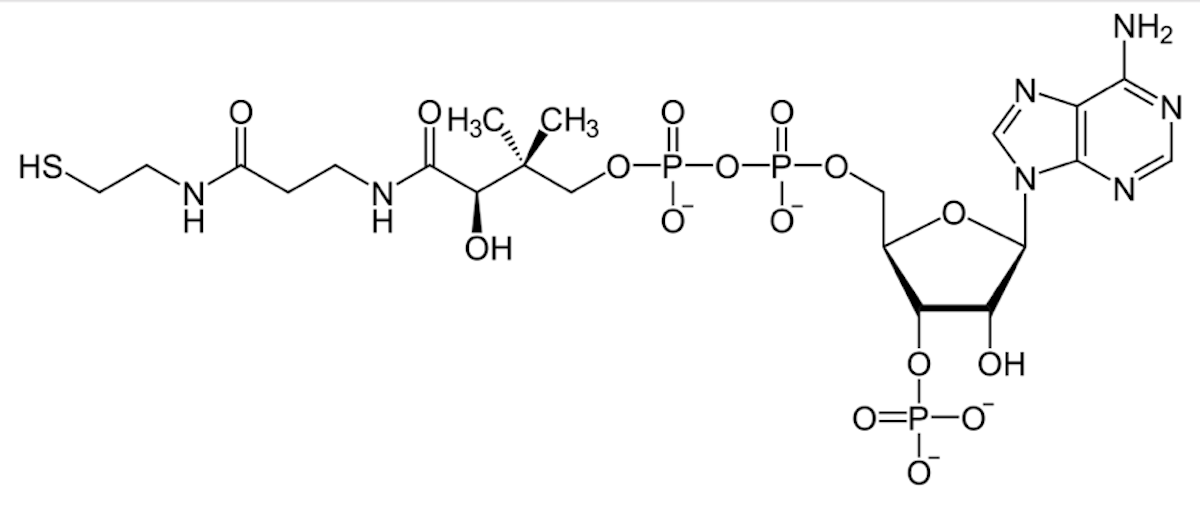
The structure of AMP, adenosine monophosphate. From Concepts of Biology – 1st Canadian Edition, Chapter 9, Figure 1, Creative Commons.
Editor’s note: See also Dr. Gauger’s earlier post, “The Hidden City.”
Biologists argue about which came first, metabolism or replication, or in other words, enzymes or DNA. Other biologists try to square the circle by saying, “We can have both at the same time with RNA.” But there is something missing from all three scenarios, without which they won’t get far. Both metabolism and replication require a means to store and transfer energy.
Modern day cells use a molecule called ATP for this process: adenosine triphosphate. It is related structurally to one of DNA’s building blocks: adenine. Compare their structures. Adenine is a base (double ring structure in orange). Add a sugar to it and it becomes adenosine. Add a phosphate to that and you have a nucleotide. Add two more phosphates, and you have ATP. Adenosine triphosphate.
“Ho Hum,” You Say
What is so interesting about that?
Think of it this way. You have a computer system that stores its information in codes of four instead of two. Not 0s and 1s, but As, Cs, Ts, and Gs. Adenine is the A. One part of the code. Now imagine you take your computer hardware that does the encoding for A, whatever it is, stick a sticky bit on it, and a rechargeable battery, and suddenly you can light campfires and suck the power from light bulbs.
This is an exaggeratedly absurd example, but it is still fascinating how the cell can reuse the same basic structural element, adenine, in several ways. It’s because of that double ring structure and all the nitrogens. They are really good at moving electrons around. And when combined with supercharged phosphate groups and the adenine in ATP, there is the potential for some serious stored energy.
Children of Adenine
Now let’s look at the other children of adenine, all important cofactors (helper molecules that perform key roles in multiple reactions in the cell).
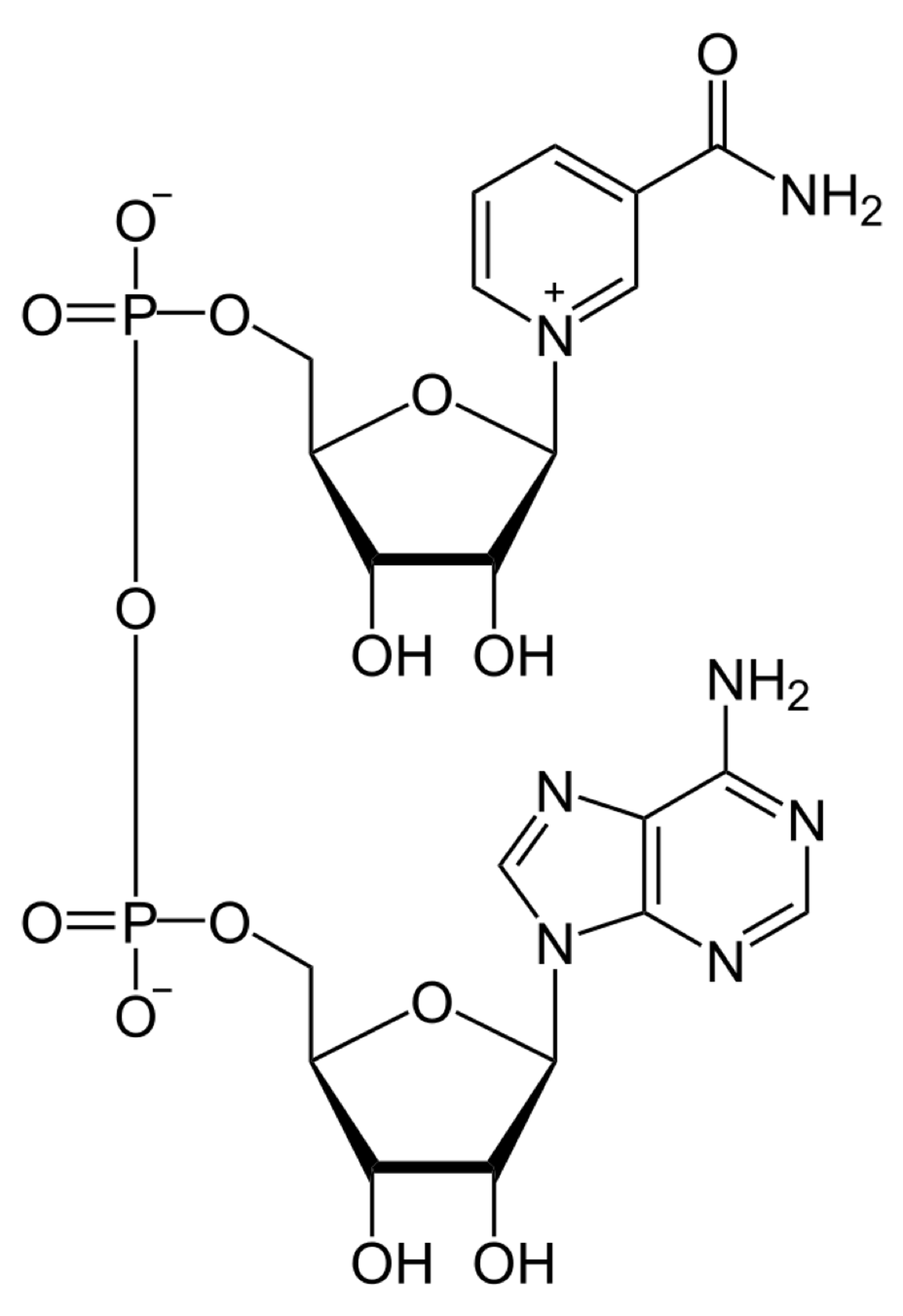
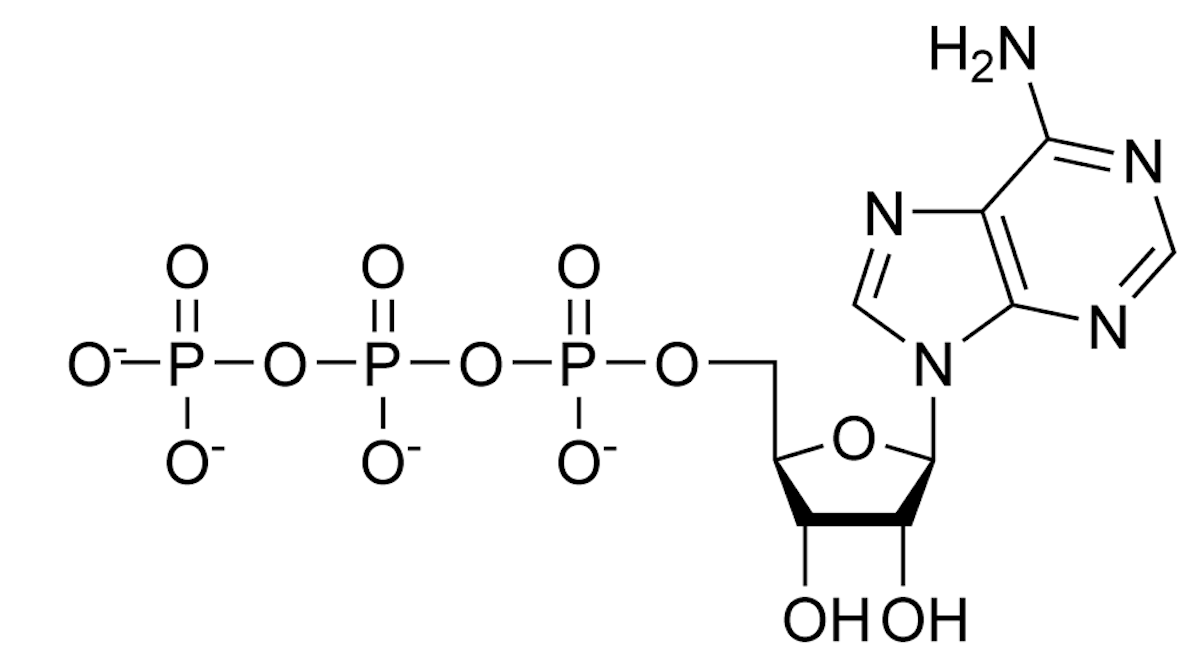
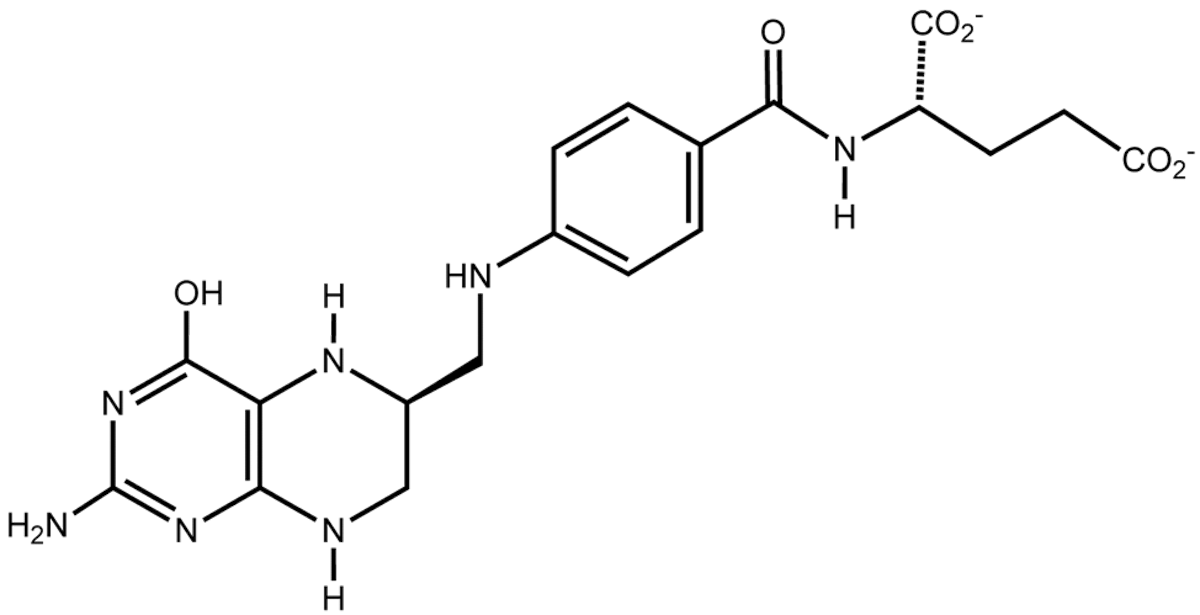
Top, NAD; below that, ATP; middle is tetrahydrofolate; and bottom is Coenzyme A. All drawings are in the public domain, by declaration of the author Neurotiker, at Wikipedia. Notice the adenine and phosphate groups in nearly every structure.
One evolutionary story is that these molecules all evolved from a common pool of compounds, using what was to hand. If you want to handle high octane electrons or shift energy of carbon groups around , then you will need help from molecules that can handle the flux. Even if they could only help part way, it would be better than nothing, and they would experiment their way to the robust, almost miraculous molecules we have today. So the story goes.
Interesting theory. Except none of these molecules can make themselves, starting from simpler molecules. They all need to be jump-started in a free-living alga that otherwise makes everything for itself.
ATP needs ATP. NAD needs NAD plus ATP. THF needs ATP, NAD, and THF. And CoA needs all four. Why?? Talk about bootstraps!
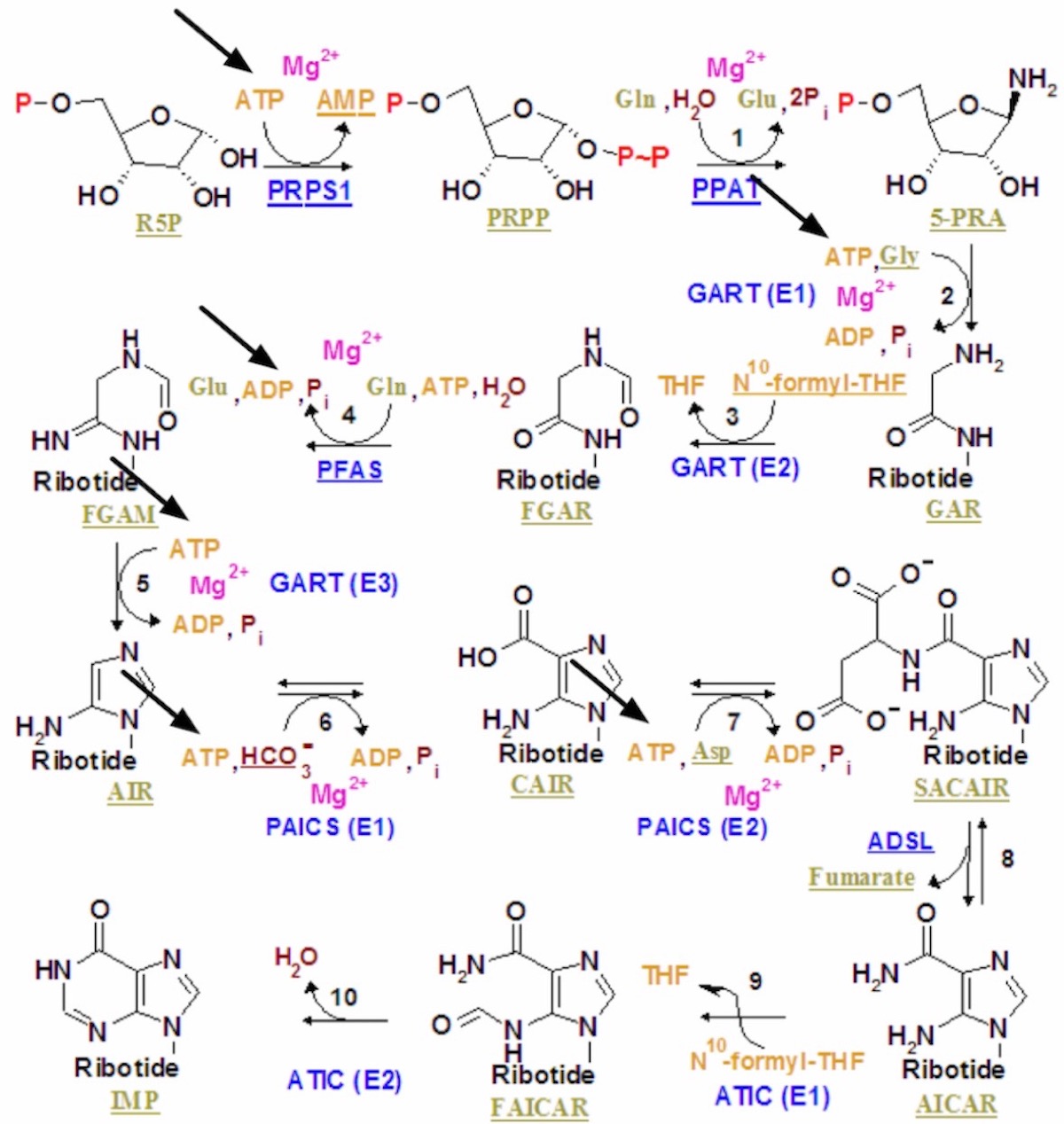
The making of IMP, from the Wikipedia page Phosphoribosylamine.
ATP is at the end of a long biosynthetic pathway to make adenine. You can’t make adenine without all these enzymes and cofactors and ATP. Multiple steps (six from ribose-5′-phosphate to IMP) along the way require ATP in order for the biosynthesis to proceed, and IMP is not yet adenine. This, it seems to me, is a show-stopper.
Assuming Nucleotides
A lot of origin-of-life stories start by assuming nucleotides. But look what it takes to get them. Unless there was an alternate genetic system that did not require nucleotides or phosphate or ATP (for energy transfer) in the very beginning, there is no way forward. RNA won’t work, because RNA is made of nucleotides too.
A Great Sucking Sound
The energy problem overshadows it all. There’s a great sucking sound of ATP disappearing down the great Atlantic rift. No way. If you need six ATP to make one ATP, no wonder you need the pump primed. More like a fire hose. There has to be another solution. The cell still needs ATP for power transfer. (Spoiler alert: There is another solution.)
Heck. How did this happen, and why? Is it universal among autotrophs (organisms that make their own food) for ATP to need itself to make itself? Can there be a metabolism with an alternate energy transfer? The answer to some of these questions may already be known. All in all, it’s a grand research question. It can’t have been by bumbling around; otherwise how did it ever produce anything like this?

From Prilifish at Flickr, red sea anemone and fish.
Cross-posted with permission from Dr. Gauger’s website, Making Notes of the Moment.
