 Evolution
Evolution
 Intelligent Design
Intelligent Design
A Mystery: Prebiotic Synthesis of Simple Organic Monomers

Editor’s note: We are delighted to present a series by Walter Bradley and Casey Luskin on the question, “Did Life First Arise by Purely Natural Means?” This is the third entry in the series, a modified excerpt from the recent book The Comprehensive Guide to Science and Faith: Exploring the Ultimate Questions About Life and the Cosmos. Find the full series so far here.
The Miller-Urey experiments were conducted in 1952–19531 and were celebrated as a great breakthrough in the search for a chemical pathway from gases assumed to be present in the early Earth’s atmosphere to chemical reactions that produced amino acids, the building blocks for protein molecules. This experiment (see the apparatus depicted below), and other similar experiments, have produced additional simple monomers — certain building blocks of life.
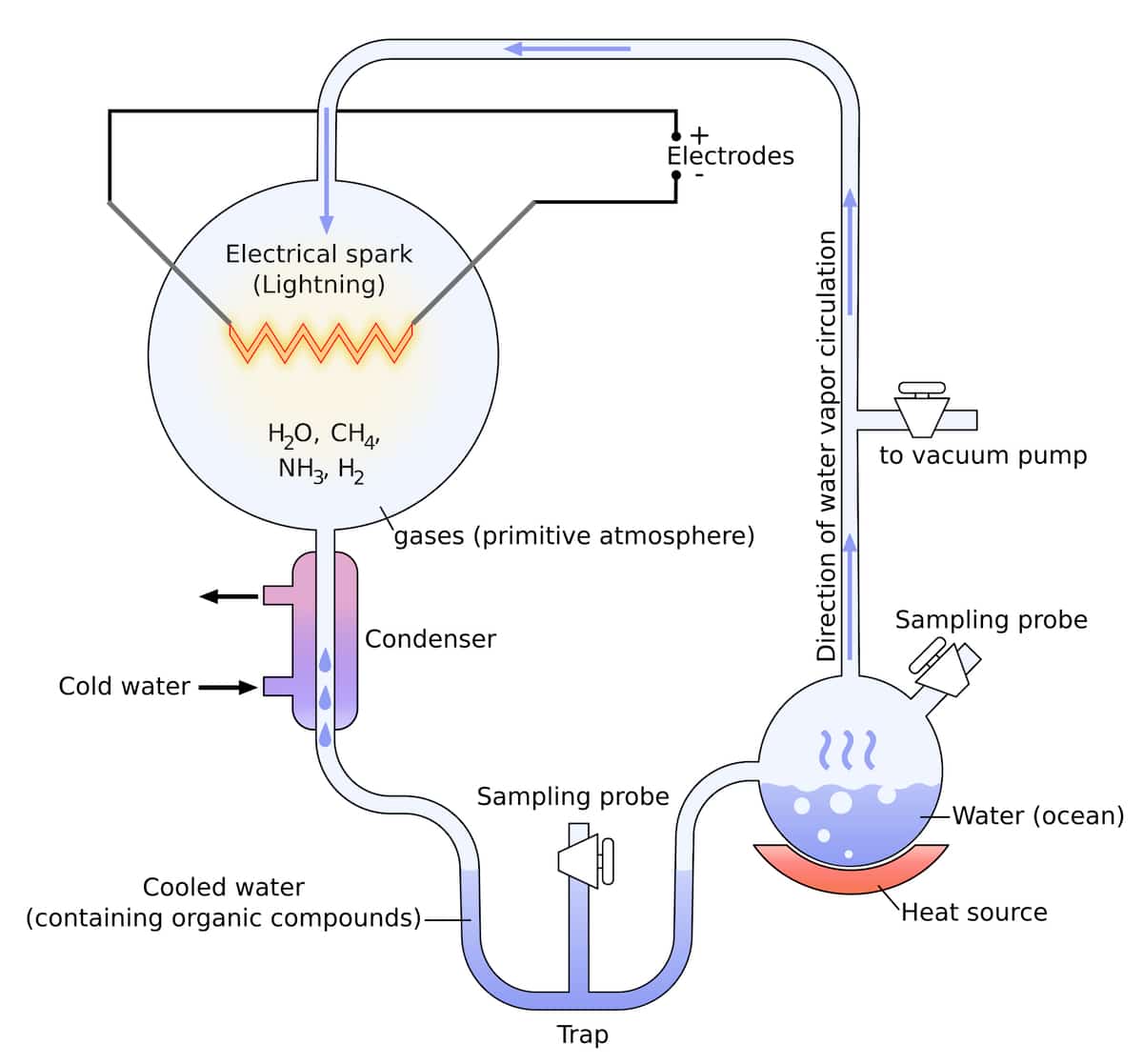
Critiques of Miller-Urey
Subsequently, careful critiques of the Miller-Urey experiments and similar experiments created great doubt about their significance, though they are still taught in some high school textbooks as if they were scientifically sound. The atmosphere used in their experiments assumed a very energy-rich primordial atmosphere of methane, ammonia, and hydrogen, none of which would have been chemically stable in an early-Earth atmosphere. Studies of the early Earth’s atmosphere by NASA during the 1980s confirmed that the mix of atmospheric gases used in the groundbreaking Miller-Urey experiments was wrong. The journal Science summed up the discoveries in 1980 by noting, “No geological or geochemical evidence collected in the last thirty years favors an energy rich, strongly reducing primitive atmosphere (i.e., hydrogen, ammonia, methane, with no oxygen). Only the success of the Miller laboratory experiments recommends it.”2 Later articles put it equally bluntly — in 1995, Science stated that “the early atmosphere looked nothing like the Miller-Urey situation.”3 Again in 2008, an article in Science reported, “Geoscientists today doubt that the primitive atmosphere had the highly reducing composition Miller used.”4
Atmosphere of the Early Earth
There are good reasons to understand why the Earth’s early atmosphere did not contain high concentrations of methane, ammonia, or other reducing gases. Earth’s early atmosphere is thought to have been produced by outgassing from volcanoes, and the composition of those volcanic gases is related to the chemical properties of the Earth’s inner mantle and core. Geochemical studies have found that the chemical properties of the Earth’s interior would have been very similar in the past as they are today.5 But today, volcanic gases do not contain methane or ammonia, and are not generally reducing. Instead, an atmosphere dominated by carbon dioxide is preferred, but this poses a problem for prebiotic synthesis experiments, as prominent origin of life theorist David Deamer observed: “Carbon dioxide does not support the rich array of synthetic pathways leading to possible monomers, so the question arose again: what was the primary source of organic carbon compounds?”6
Another problem with Miller-Urey type prebiotic synthesis experiments is that when amino acids are synthesized from energy-rich gases, a racemic mixture of amino acids is created with 50 percent L-amino acids and 50 percent D-amino acids, sometimes called left-handed and right-handed. Proteins molecules created in living systems must have 100 percent L-amino acids. If there are any D-amino acids in the chain, it would prevent the chain of amino acids from folding up into the proper three-dimensional protein structures associated with this amino acid string, preventing it from performing its function.
More Problems with Miller-Urey
There are many additional problems with Miller-Urey-type research that seeks to identify plausible chemical pathways for the synthesis of proteins, DNA, and RNA molecules — the molecules of life. So drastic is the evidence against prebiotic synthesis of life’s building blocks that in 1990, the Space Studies Board of the National Research Council recommended a “reexamination of biological monomer synthesis under primitive Earthlike environments, as revealed in current models of the early Earth.”7 Because of these difficulties, many leading theorists have abandoned the Miller-Urey experiment and the “primordial soup” model it is claimed to support. In 2010, University College London biochemist Nick Lane stated the primordial soup theory “doesn’t hold water” and is “past its expiration date.”8 Instead, he proposes that life arose in undersea hydrothermal vents where water circulates through hot volcanic rock at the bottom of the ocean. But both the hydrothermal vent and primordial soup hypotheses face another major problem.
Next, “Forming Polymers: A Problem for the Origin of Life.”
Notes
- Stanley L. Miller, “A Production of Amino Acids Under Possible Primitive Earth Conditions,” Science 117 (May 15, 1953), 528-529.
- Richard A. Kerr, “Origin of Life: New Ingredients Suggested,” Science 210 (October 3, 1980), 42-43.
- Jon Cohen, “Novel Center Seeks to Add Spark to Origins of Life,” Science 270: 1925-1926 (December 22, 1995).
- Adam P. Johnson, “The Miller Volcanic Spark Discharge Experiment,” Science 322 (October 17, 2008), 404.
- Kevin Zahnle, Laura Schaefer, and Bruce Fegley, “Earth’s Earliest Atmospheres,” Cold Spring Harbor Perspectives in Biology 2(10), a004895 (October 2010) (“Geochemical evidence in Earth’s oldest igneous rocks indicates that the redox state of the Earth’s mantle has not changed over the past 3.8 Gyr”); Dante Canil, “Vanadian in peridotites, mantle redox and tectonic environments: Archean to present,” Earth and Planetary Science Letters 195:75-90 (2002).
- David W. Deamer, “The First Living Systems: a Bioenergetic Perspective,” Microbiology & Molecular Biology Reviews 61:239 (1997).
- National Research Council Space Studies Board, The Search for Life’s Origins (Washington, DC: National Academy Press, 1990).
- Deborah Kelley, “Is It Time to Throw Out ‘Primordial Soup’ Theory?,” NPR (February 7, 2010).
