 Intelligent Design
Intelligent Design
Inside the Machine Room of the Nucleus
We’re living in exciting times. It was only a little over 65 years ago that we began to realize that life is information-based, with coded genetic information embedded in alphabet-like molecules. In the years that followed, biochemists also began to discover that the chemical reactions carried on by life were unlike the valence-based reactions we learned about in chemistry class, but were machine-like, with moving parts that function mechanically like rotors and engines. More recently, these machines have been found to interact in large networks of integrated systems, like factories. Is it any wonder that intelligent design rose as a movement in the late 20th century?
The wonders continue at a rapid pace into the 21st century. With increasing resolution thanks to techniques like cryo-electron microscopy, biochemists are focusing on the molecular machines at nearly nanometer scale and determining their modes of action. Let’s look at news about a few of the major players inside the nucleus of cells.
The Majestic Spliceosome
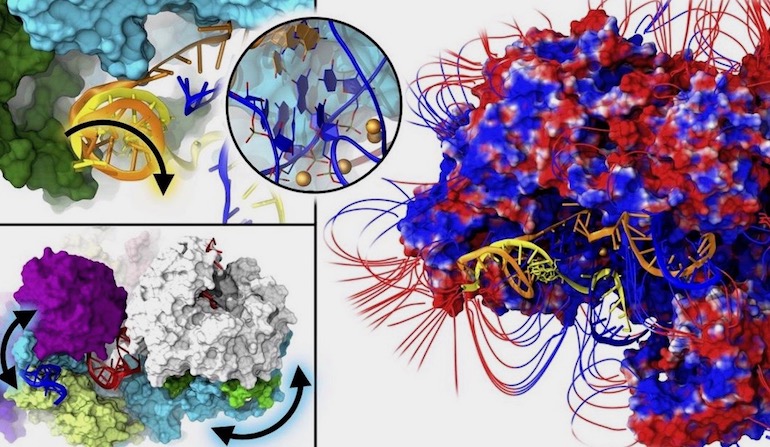
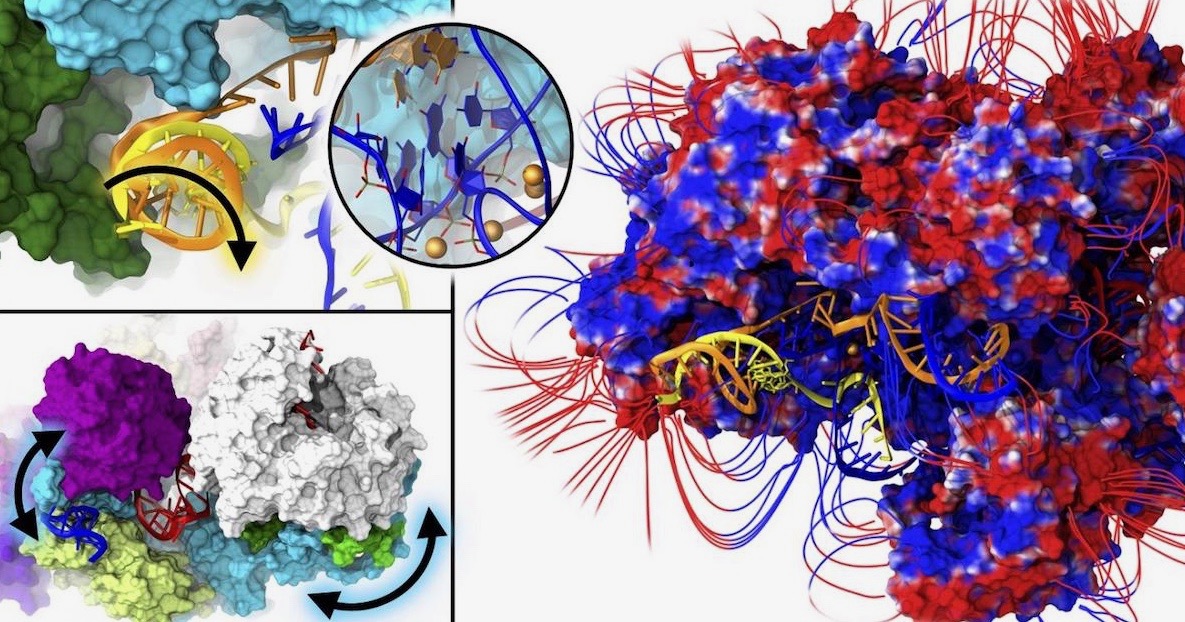
Researchers at the International School of Advanced Studies (SISSA) in Italy use a rare word in a scientific paper: “majestic.” It’s an appropriate adjective for what they saw by examining, for the first time, the spliceosome at near-atomic detail. Their paper in PNAS begins:
The spliceosome (SPL) is a majestic macromolecular machinery composed of five small nuclear RNAs and hundreds of proteins. SPL removes noncoding introns from precursor messenger RNAs (pre-mRNAs) and ligates coding exons, giving rise to functional mRNAs. Building on the first SPL structure solved at near–atomic-level resolution, here we elucidate the functional dynamics of the intron lariat spliceosome (ILS) complex through multi-microsecond-long molecular-dynamics simulations of ∼1,000,000 atoms models. The ILS essential dynamics unveils (i) the leading role of the Spp42 protein, which heads the gene maturation by tuning the motions of distinct SPL components, and (ii) the critical participation of the Cwf19 protein in displacing the intron lariat/U2 branch helix. These findings provide unprecedented details on the SPL functional dynamics, thus contributing to move a step forward toward a thorough understanding of eukaryotic pre-mRNA splicing. [Emphasis added.]
One paper could never do justice to all that the spliceosome does, but in their examination of a couple of protein parts, they could not help but stand in awe at the majesty of this huge molecular machine. They didn’t have much to say about evolution, except pointing out that one “intricate RNA-based active site has been extraordinarily conserved across evolution from bacteria to humans.” News from SISSA via Phys.org recaps the awe by showing the precision the team found in this machine’s operation:
To have a molecule able to transport information usefully, precisely and effectively, the introns must be eliminated by the spliceosome to transform it into mature mRNA.” It is an extremely precise snip-and-stitch process, explains the researcher, because any tiny error can alter the information with serious effects on cell activity and the health of the entire organism.
Topoisomerase the Great
“Supercoil me!” begins the headline of another article from SISSA about “the art of knotted DNA maintenance” (see Science Daily). Is it any surprise that long strands of DNA have knotty problems? Like those old phone cords that would knot up with coils upon coils, DNA can develop messy knots. Specialized enzymes must untangle them; otherwise, important genes might become inaccessible. Like Alexander the Great cutting the Gordian knot, they say, the topoisomerase family of nuclear machines is up to the challenge. They get some help, though, by a physical property of DNA that works to the advantage of the cutting machines: “Supercoiling can keep DNA knots locked in place for long enough that they can be untied by specialised enzymes,” the new study shows.
“We know that living cells routinely deal with DNA knots, and we also know that these forms of entanglement are usually detrimental to biological functionality; for instance they can prevent the genetic information from being read and translated into protein products. Specific enzymes of the topoisomerase family are responsible for disentangling DNA. Their modus operandi recalls the efficient, if not drastic, way in which Alexander the Great untied the proverbial Gordian knot with a cut of his sword. Similarly, these enzymes untie DNA filaments by a sophisticated cutting and sealing action.”
The machines have another problem not faced by Alexander: DNA strands are constantly wiggling. Using computer simulations, the team found that free knots would slide relatively fast along the filaments. This should make it more difficult for the topoisomerases to get to them and repair them. The team then noticed that accidental knots are often associated with supercoiled regions, which are more stable. This led to an “Aha!” moment:
Our study suggests that DNA supercoiling can favour the action of topoisomerases by keeping knots in a stable configuration for a time span that is much longer than other molecular rearrangements. In this way, the enzymes could have sufficient time to recognise the target sites and, in turn, their cut-and-paste action would be simpler, more reliable and efficient.
Their hypothesis will require further testing, but suggests an additional purpose for supercoiling inside the nucleus. It was already known that supercoiling is involved in “exposing or hiding genetic information” for transcription, but now another possibility presents itself: supercoils hold an accidental knot in place long enough for Topoisomerase the Great to arrive with his sword.
“We studied the DNA filament and noted that without supercoils, the knotted region would move relatively fast along the filament. Likewise, supercoiled regions can rapidly change in knot-free DNA. However, when knots and supercoiling are simultaneously present, then the crucial contact points in DNA knots become locked in place, persistently. And this unexpected effect is particularly interesting because it could be key to a specific and unexpected biological functionality.”
It will be interesting to find out if other machines notice the knots and induce supercoiling at those locations. Sounds like a good plan at least.
Scalpel in a Sheath
For our last example of nuclear machines today, consider 53BP1. This enzyme has a critical job: repairing double-stranded breaks, when both strands of DNA are broken. It’s a catastrophic condition leading to cell death. Fortunately, cells come equipped with machinery that can stitch them back together. One of them is 53BP1. But just as you don’t want open knives floating around, you only want this machine activated when it is needed. Fortunately again, this machine comes with a sheath named TIFF. News from the Mayo Clinic explains (you can ignore the evolutionary claim in the first sentence):
Damage to DNA is a daily occurrence but one that human cells have evolved to manage. Now, in a new paper published in Nature Structural & Molecular Biology, Mayo researchers have determined how one DNA repair protein gets to the site of DNA damage….
While the human genome is constantly damaged, cells have proteins that detect and repair the damage. One of those proteins is called 53BP1. It is involved in the repair of DNA when both strands break.
Dr. Georges Mer and team found that TIFF normally binds to 53BP1, rendering it harmless like a sword in its sheath.
The authors report that when DNA damage occurs, RNA molecules produced at that time can bind to TIRR, displacing it from 53BP1 and allowing 53BP1 to swing into action.
Presumably the binding is reversible after the repairs are made, and the Swiss army knife goes back into the pocket till next time. Pretty cool, heh? Read more about the machine in Nature Reviews Molecular Cell Biology (2014).
These are brief looks at the amazing machines and processes that keep us going without our conscious awareness. When you ponder the fact that until our day nobody knew about these wonders, our reasons for feeling like a Privileged Species in the 21st century take on historic significance.
Image: Closer look at the spliceosome, by Alessandra Magistrato, via Eurekalert!
